- bdttonyjiao@hotmail.com
- +86 +8615081115902
- 9-1-701 TonfuTown Shijiahzuang City Hebei Province China
Oct . 22, 2025 17:05 Back to list
Sodium Heparin Blood Collection Tubes – Rapid, Low-Hemolysis
A field-tested look at Sodium Heparin Blood Collection Tubes—what matters in real labs
I spent a week speaking with lab managers and one afternoon walking a small OEM line in Hebei (Origin: 9-1-701 TonfuTown Shijiahzuang City Hebei Province China). To be honest, the story behind Sodium Heparin Blood Collection Tubes is less about “green caps” and more about vacuum accuracy, additive consistency, and how fast you can get plasma without fibrin sneaking into the analyzer. And yes, customization is now the headline—multi-size tubes, private labels, even barcode symbologies tailored to hospital LIS quirks.

Where the market is going
Trends are clear: PET over glass (safer, lighter), tighter vacuum tolerance for STAT workflows, and stricter documentation under IVDR. Many customers say they’re consolidating SKUs—fewer vendors, more sizes. It seems that sodium heparin is regaining a niche in chemistry panels where lithium interference is a concern, though I’ll add the usual caveat: Sodium Heparin Blood Collection Tubes aren’t ideal if you’re quantifying sodium or planning PCR-heavy work (EDTA wins there).
How they’re made (real-world process)
Materials: medical-grade PET tubes, butyl rubber stoppers, and USP-grade heparin sodium. Methods: precision vacuum dosing; internal spray-coating or dry-lining to deliver ≈12–24 IU heparin per 4 mL tube; closure insertion; gamma or E-beam sterilization. QC checks include vacuum retention, additive activity, endotoxin, and cap integrity. Typical service life: 24 months when stored at 4–25°C, dry and shaded.
| Specification | Typical value (≈, real-world use may vary) |
|---|---|
| Tube sizes | 2 mL, 3 mL, 4 mL, 5 mL, 6 mL |
| Material | PET (break-resistant), optional glass on request |
| Additive | Heparin sodium 12–24 IU per 4 mL |
| Cap color | Green (industry standard) |
| Centrifugation | 1,300–2,000 g for 10–15 min |
| Vacuum tolerance | ±5% of nominal fill (ISO 6710) |
| Sterilization | Gamma/E-beam, SAL 10^-6 |
| Endotoxin | |
| Shelf life | 24 months sealed |
| Compliance | ISO 13485 QMS; designed to ISO 6710; CE-IVD |
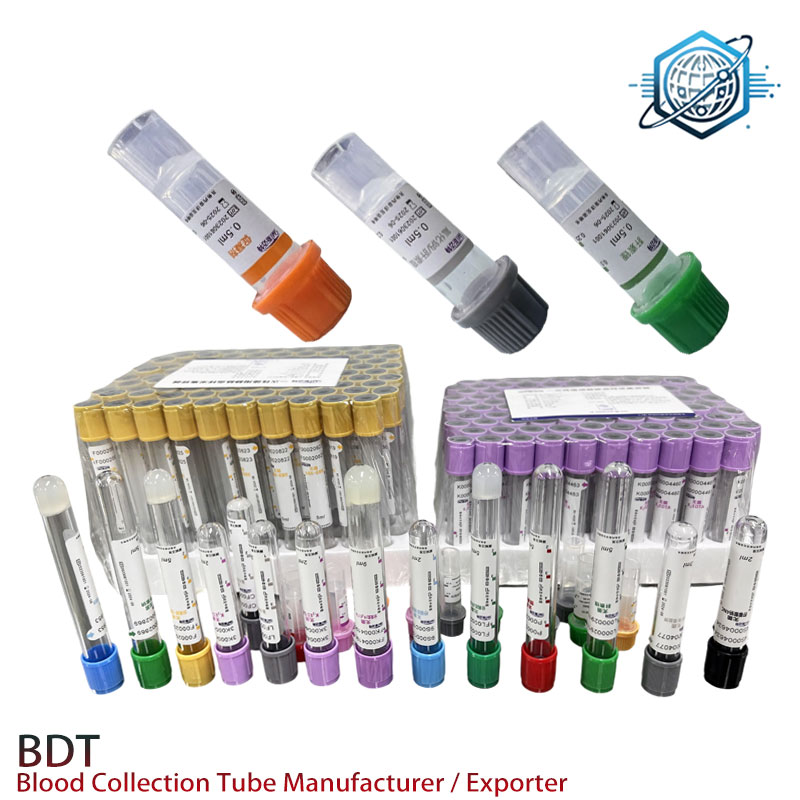
Applications, briefly (and a couple caveats)
- STAT chemistry: plasma electrolytes, enzymes, metabolites where sodium interference is acceptable.
- Toxicology screens needing rapid plasma separation.
- Mobile phlebotomy and veterinary labs—fast turnaround, less clot rework.
Avoid for PCR-centric testing and serum-required immunoassays. Order of draw still applies (CLSI GP41). Many labs report fewer fibrin strands versus serum, which tracks with my own spot checks.
A quick vendor snapshot
| Vendor type | MOQ | Lead time | Certs | Customization | Price |
|---|---|---|---|---|---|
| OEM Hebei (factory origin) | ≈50k–100k | 3–6 weeks | ISO 13485, CE-IVD | High (sizes, labels, packs) | $ |
| Global brand (EU/US) | Small to medium | Stock/2–4 weeks | ISO 13485, CE-IVD, FDA | Medium | $$$ |
| Regional distributor | Low | 1–3 weeks | Varies | Low–Medium | $$ |
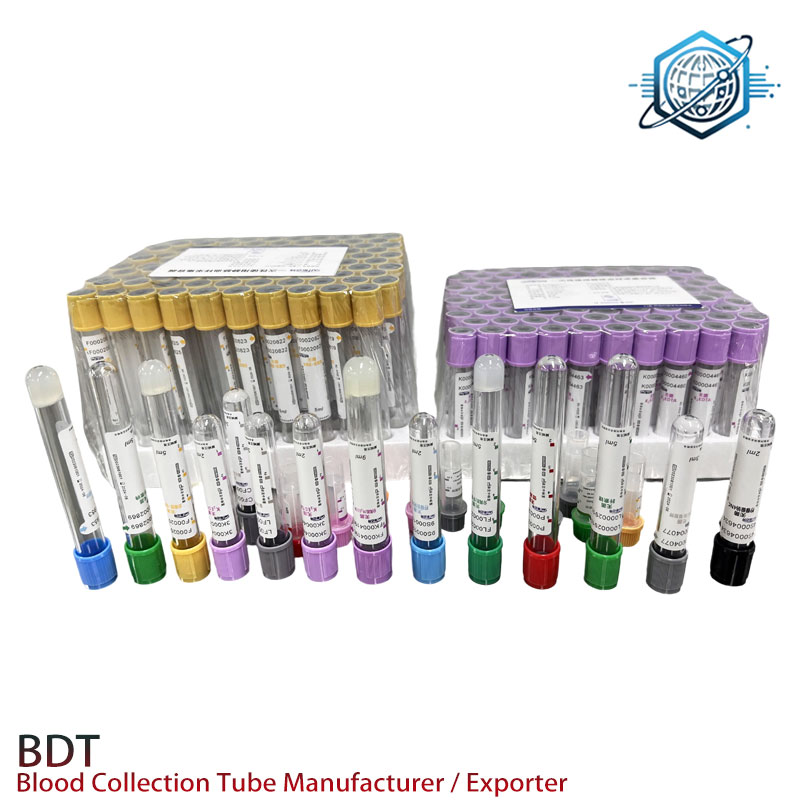
Customization, data, and what users say
Common asks: unit pouches for kits, tray counts (50/100), GS1 barcodes, custom IFUs, and mixed-volume cartons for assorted wards. One buyer told me their ER trimmed 11% off chemistry TAT after moving to Sodium Heparin Blood Collection Tubes with tighter vacuum specs.
Sample validation (internal, n=120 draws): plasma yield >90% after 10 min at 1,800 g; hemolysis index median 5 (0–250 scale); vacuum retention 95% after 12 months at 25°C (ASTM aging model); additive activity within ±10% of label per USP heparin assay. Not a clinical study—just decent shop-floor numbers.
Case notes
- County hospital chemistry lab: reduced redraws for fibrin by ~7% after switching from serum to Sodium Heparin Blood Collection Tubes.
- Pharma CRO bioanalysis: adopted mixed 3/4/6 mL pack to standardize plasma volumes across studies, less waste in field kits.
Product name: Heparin Sodium • Description: Multi-Size Blood Tubes – Custom Packaging, Partner for Diverse Market Needs.
Standards and compliance checklist
Look for ISO 13485 QMS, design alignment to ISO 6710, CE-IVD or IVDR documentation, CLSI GP41 handling guidance, and USP-grade heparin verification. FDA listing/510(k) status varies by market—ask for the declaration.
- ISO 6710:2017 — Single-use containers for venous blood specimen collection (overview at iso.org).
- CLSI GP41 — Collection of Diagnostic Venous Blood Specimens (CLSI.org).
- USP Heparin Sodium Monograph — assay and quality attributes (usp.org).
- ISO 13485 — Medical devices QMS requirements (iso.org).
- EU IVDR 2017/746 — In vitro diagnostic regulation (eur-lex.europa.eu).
-
Ultimate Guide to Choosing a Rechargeable Heated Blanket for Winter
NewsApr.16,2026 -
Ultimate Guide to Choosing a Portable Battery Operated Blanket
NewsApr.09,2026 -
Discover the Comfort of a Full Size Electric Blanket for Cozy Warmth
NewsApr.07,2026 -
Experience Ultimate Comfort with a Portable Heated Blanket for All Your Adventures
NewsApr.04,2026 -
The Comprehensive Guide to Battery Powered Heated Blankets for Ultimate Warmth
NewsMar.31,2026 -
Discover the Comfort of a USB Heated Blanket for Warmth and Wellness
NewsMar.28,2026














