- bdttonyjiao@hotmail.com
- +86 +8615081115902
- 9-1-701 TonfuTown Shijiahzuang City Hebei Province China
Sep . 02, 2025 05:00 Back to list
K EDTA Tube: Premium Blood Collection for Accurate Lab Results
Introduction to Advanced Blood Collection Technologies
In the sophisticated realm of clinical diagnostics and research, the integrity of blood samples is paramount for accurate results. The choice of blood collection tubes significantly impacts sample quality, influencing everything from hematology to molecular biology assays. Among the various types, the k edta tube stands out as a critical component, primarily utilized for whole blood collection in hematological analysis. Ethylenediaminetetraacetic acid (EDTA) acts as an anticoagulant by chelating calcium ions, thereby preventing blood coagulation without altering cellular morphology, which is crucial for complete blood count (CBC) and blood typing. This article delves into the technical aspects, manufacturing precision, and diverse applications of these essential laboratory consumables, providing a comprehensive overview for B2B decision-makers and technical professionals.
The demand for high-quality k edta tubes is steadily growing, driven by advancements in diagnostic techniques and the increasing volume of laboratory tests globally. Modern clinical laboratories require tubes that not only meet stringent anticoagulant efficacy standards but also offer superior material compatibility, reduced risk of hemolysis, and precise volume accuracy. Our focus here is to highlight how superior manufacturing processes and adherence to international quality standards contribute to the reliability and performance of these vital tools in medical diagnostics.
The Precision Manufacturing Process of the K2 EDTA Tube
The production of a high-quality k edta tube is a highly technical and controlled process, ensuring product integrity and consistent performance. This process integrates advanced engineering with meticulous quality control, reflecting the critical nature of its application in patient diagnostics. Below is a detailed breakdown of the manufacturing flow, emphasizing the materials, processes, and stringent testing protocols.
Key Materials and Components:
- Tube Body: Typically medical-grade Polyethylene Terephthalate (PET) or polypropylene (PP), selected for its clarity, chemical inertness, and gas barrier properties. PET is preferred for its vacuum retention capabilities.
- Stopper: High-quality butyl rubber, offering excellent sealing, puncture resistance, and re-sealing properties to prevent evaporation and contamination.
- Anticoagulant: Dipotassium EDTA (K2EDTA) or Tripotassium EDTA (K3EDTA), precisely sprayed on the inner wall of the tube to ensure optimal blood-to-anticoagulant ratio.
- Cap: Made from PP, designed for secure sealing and easy opening.
Manufacturing Process Flow:
Tube Molding (Casting/Injection Molding): Medical-grade PET resin is melted and injection-molded into precision tube bodies. This ensures consistent dimensions and wall thickness crucial for vacuum integrity.
EDTA Spray Drying: A precise quantity of K2EDTA solution is atomized and sprayed uniformly onto the inner surface of the tubes. This is a critical step, as the concentration and even distribution of EDTA directly impact its anticoagulant efficacy. The tubes are then dried under controlled conditions.
Vacuum Formation: The tubes are evacuated to a precise vacuum level. This step is crucial for drawing the correct volume of blood during collection. Advanced vacuum forming techniques ensure long-term vacuum stability.
Capping and Sealing: Butyl rubber stoppers and plastic caps are automatically inserted and crimped, ensuring an airtight seal and tamper-evidence. This maintains the vacuum and sterility of the tube.
Sterilization: The assembled tubes undergo terminal sterilization, typically by gamma irradiation, to achieve a Sterility Assurance Level (SAL) of 10^-6, in accordance with ISO 11137 standards.
Quality Control and Packaging: Each batch undergoes rigorous testing for vacuum integrity, anticoagulant efficacy, sterility, and dimensional accuracy. Finished products are labeled and packaged in cleanroom environments.
Testing Standards and Service Life:
Our k edta tubes adhere strictly to international standards such as ISO 6710 (Single-use container111s for human venous blood specimen collection) and CLSI H3-A6 (Procedures for the Collection of Diagnostic Blood Specimens by Venipuncture). These standards dictate critical parameters including fill volume accuracy, anticoagulant concentration, vacuum retention, and material biocompatibility. The typical service life of an unopened, properly stored EDTA tube is 12-24 months from the date of manufacture, maintaining its vacuum and anticoagulant properties.
Target Industries and Application Advantages:
The primary target industries for k2edta blood collection tubes are clinical diagnostics laboratories, hospitals, blood banks, and research institutions. In these settings, the tubes demonstrate critical advantages:
- Reliable Anticoagulation: Ensures accurate hematological parameters for CBC, ESR, and blood smear evaluations.
- Reduced Hemolysis: Optimized tube materials and anticoagulant application minimize cellular damage, preserving sample integrity.
- Enhanced Workflow Efficiency: Pre-measured anticoagulant and consistent vacuum allow for quick, standardized blood collection.

Technical Specifications and Performance Parameters
Understanding the detailed technical specifications of k edta tubes is essential for laboratories to ensure compatibility with their analytical equipment and to guarantee the accuracy of diagnostic results. Our EDTAK2 tubes are engineered to meet the highest industry standards, providing reliable performance for a wide array of hematological tests.
EDTAK2 Product Specification Table:
The selection of K2EDTA over K3EDTA is a deliberate decision based on current clinical guidelines. Studies suggest that K2EDTA has less impact on red blood cell morphology and volume, particularly when samples are stored for extended periods, making it the preferred anticoagulant for hematology analysis and maintaining the stability of cellular components. This is a crucial consideration for k2 edta tube use in modern diagnostic protocols.
Application Scenarios and Real-World Case Studies
The versatility and reliability of k edta vacutainer tubes make them indispensable across a broad spectrum of medical and research applications. Their primary role is in collecting whole blood specimens where the cellular components need to be preserved without coagulation for subsequent analysis.
Typical Application Areas:
- Hematology: Complete Blood Count (CBC), erythrocyte sedimentation rate (ESR), reticulocyte count, platelet count, and blood smear morphology analysis.
- Blood Banking: Blood typing and cross-matching, ensuring compatibility for transfusions.
- Molecular Diagnostics: Isolation of DNA from whole blood for genetic testing, PCR-based assays, and other molecular analyses where cellular integrity is key.
- Immunology: Flow cytometry for immunophenotyping, allowing the analysis of various cell populations.
Application Case Study: Large Scale Hematology Screening Program
Client: A leading national public health laboratory in Southeast Asia, conducting routine hematology screenings for millions of citizens annually.
Challenge: The client required a consistent supply of high-quality blood collection tubes that could withstand high-throughput automated processing without compromising sample integrity. They had previously experienced issues with inconsistent vacuum, which led to underfilling, and variable EDTA concentration, affecting CBC results.
Solution & Impact: By transitioning to our EDTAK2 tubes, the laboratory observed a significant improvement in sample quality and processing efficiency. The precise vacuum control ensured consistent fill volumes, reducing the need for recollection. The uniform K2EDTA coating prevented microscopic clots, which can interfere with automated analyzers and cause erroneous low platelet counts.
"Switching to EDTAK2 tubes dramatically reduced our sample rejection rates due to clotting or insufficient volume. Our technicians report improved ease of use, and our hematology analyzers consistently provide more accurate results, which is vital for our public health initiatives."
— Dr. Anya Sharma, Head of Hematology, National Public Health Lab
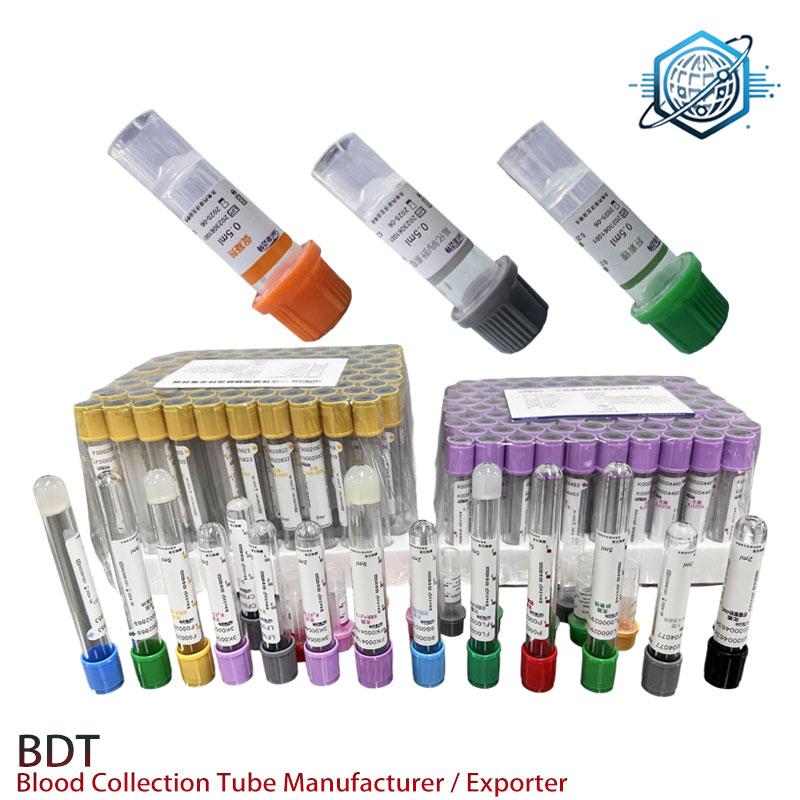
This case highlights the direct impact of superior product design and manufacturing on clinical outcomes and operational efficiency. The consistent performance of our k2 edta vacutainer tubes ensures laboratories can focus on accurate diagnostics rather than troubleshooting sample integrity issues.
Technical Advantages and Performance Benchmarks
The technical superiority of our EDTAK2 tubes is rooted in meticulous design and adherence to stringent quality controls, offering distinct advantages that elevate diagnostic reliability and laboratory efficiency.
- Optimized K2EDTA Concentration: Our tubes feature an optimized concentration of K2EDTA (1.8 mg/mL blood), precisely sprayed to ensure rapid and complete anticoagulation without diluting the sample or affecting cellular morphology. This reduces the risk of platelet aggregation and microclot formation, common issues with less precise applications.
- Superior Vacuum Stability: Engineered with advanced sealing technology and high-grade PET, our tubes maintain vacuum integrity for their entire shelf life. This guarantees accurate fill volumes, preventing underfilling errors that can lead to incorrect anticoagulant-to-blood ratios and compromised results.
- Robust Material Construction: The medical-grade PET plastic provides exceptional clarity for visual inspection and is shatter-resistant, enhancing safety for laboratory personnel. The butyl rubber stopper offers superior re-sealing capabilities after needle puncture, minimizing aerosol generation and leakage during transport or processing.
- Reduced Additive Interference: The K2EDTA formulation is designed to be free from heavy metals and other impurities that could interfere with downstream assays, ensuring clean and reliable results for sensitive molecular and immunological tests.
- Low Hemolysis Rate: Through careful selection of tube materials and manufacturing processes, our tubes exhibit an exceptionally low hemolysis rate, preserving the integrity of red blood cells—a critical factor for accurate hematology parameters.
- Compatibility with Automated Systems: Designed with standardized dimensions (e.g., 13x75mm, 13x100mm, 16x100mm), our k edta tubes are fully compatible with leading automated blood analysis systems, ensuring seamless integration into modern laboratory workflows and minimizing manual handling.
These advantages culminate in a product that not only meets but often exceeds industry benchmarks for safety, reliability, and diagnostic accuracy, thereby contributing significantly to efficient and trustworthy laboratory operations.
Vendor Comparison and Product Differentiation
The market for blood collection tubes is competitive, with several manufacturers offering various options. Understanding the nuances of product differentiation is crucial for procurement managers and laboratory directors when evaluating suppliers for k2 edta tan top tube or lavender top variants. Our EDTAK2 tubes are distinguished by a commitment to consistent quality, innovative manufacturing, and superior performance.
Key Differentiation Factors:
- K2EDTA Purity and Uniformity: We utilize high-purity K2EDTA and employ a sophisticated spray-drying technique that ensures an exceptionally uniform coating. This prevents localized high concentrations that could cause cell crenation or low concentrations that could lead to microclots. Competitors often use less precise methods, resulting in inconsistent anticoagulant distribution.
- Vacuum Precision and Longevity: Our manufacturing process includes advanced vacuum control systems, guaranteeing that each tube has a precise and stable vacuum, minimizing underfilling or overfilling. Many generic brands may suffer from vacuum loss over time, impacting sample volumes and test accuracy.
- Trace Element Contamination: EDTAK2 tubes are manufactured in cleanroom environments with rigorous control over potential trace element contamination. This is especially important for specialized tests where even minute levels of interfering substances can skew results. Some lower-cost alternatives may introduce unacceptable levels of trace elements.
- Material Quality and Safety: We use only premium medical-grade PET plastic and butyl rubber stoppers, chosen for their inertness, strength, and barrier properties. This ensures sample integrity and prevents leachables that could interfere with analysis. Lower-grade plastics can sometimes interact with blood components or be more prone to breakage.
Comparative Analysis Table: EDTAK2 vs. Generic K2EDTA Tubes
Choosing EDTAK2 is an investment in diagnostic accuracy and patient safety. Our differentiation lies in our unwavering commitment to precision engineering and quality assurance at every stage of production for our k edta tube products.
Customized Solutions and Services
Recognizing that every laboratory and healthcare system has unique requirements, we offer comprehensive customized solutions for our k edta tubes. Our goal is to seamlessly integrate our products into your specific operational workflows, optimizing efficiency and cost-effectiveness without compromising quality.
Tailored Customization Options:
- Specific Tube Dimensions and Fill Volumes: While standard sizes (13x75mm, 13x100mm, 16x100mm) and fill volumes (2-9mL) are available, we can produce custom dimensions or unique fill volumes to match specialized equipment or research protocols.
- Private Labeling and Branding: For large distributors or hospital networks, we offer private labeling services, incorporating your brand logo, specific color codes, and product information directly onto the tubes and packaging.
- Packaging Configurations: From bulk sterile packaging to custom-counted racks and trays, we can adapt packaging to suit automated dispensing systems or specific storage requirements.
- Anticoagulant Variations: While K2EDTA is standard, we can discuss specific needs for K3EDTA if required for particular applications, ensuring the exact anticoagulant performance needed.
- Regulatory and Documentation Support: We provide comprehensive documentation support, including Certificates of Analysis, regulatory clearances (e.g., CE, FDA 510(k) status), and technical data sheets tailored to your submission requirements.
Our dedicated team of technical experts collaborates closely with clients from initial consultation through design, prototyping, and final production, ensuring that all customized solutions meet stringent quality standards and performance criteria. We prioritize a responsive and flexible approach to customer needs, empowering our partners with products perfectly aligned with their operational demands.

Ensuring Quality: Certifications and Trust
Trust is the cornerstone of B2B relationships, particularly in the medical device sector. Our commitment to excellence is underpinned by robust quality management systems and adherence to global regulatory standards.
Authoritativeness & Certifications:
- ISO 13485 Certified: Our manufacturing facilities operate under an ISO 13485 certified Quality Management System, specifically designed for medical device manufacturing. This ensures traceability, risk management, and continuous improvement across all production stages.
- CE Mark: All our k edta tube products bear the CE Mark, indicating compliance with the European In Vitro Diagnostic Medical Device Regulation (IVDR 2017/746), confirming they meet essential health and safety requirements.
- FDA Registered: Our products are registered with the U.S. Food and Drug Administration (FDA), demonstrating compliance with relevant regulations for distribution in the United States.
- CLSI and ISO Standards: We rigorously follow CLSI H3-A6 (Procedures for the Collection of Diagnostic Blood Specimens by Venipuncture) and ISO 6710 (Single-use container111s for human venous blood specimen collection) guidelines for product design and performance testing.
Years of Service and Partner Clients:
With over 15 years of dedicated service in the medical consumables industry, we have cultivated long-standing partnerships with major hospital networks, national public health laboratories, and leading diagnostic distributors across North America, Europe, and Asia. Our track record reflects a consistent delivery of reliable, high-performance products and exceptional customer service.
Frequently Asked Questions (FAQ)
-
Q: What is the primary use of K2EDTA tubes?
A: K2EDTA tubes are primarily used for whole blood collection in hematological analysis, including Complete Blood Count (CBC), blood typing, and molecular diagnostic tests where cellular integrity is critical.
-
Q: What is the difference between K2EDTA and K3EDTA?
A: K2EDTA (Dipotassium EDTA) and K3EDTA (Tripotassium EDTA) are both effective anticoagulants. K2EDTA is generally preferred by CLSI (Clinical and Laboratory Standards Institute) due to its minimal impact on red blood cell morphology and volume, especially during prolonged storage, leading to more accurate results in hematology.
-
Q: How should EDTAK2 tubes be stored?
A: Unopened EDTAK2 tubes should be stored at room temperature (4-25°C), away from direct sunlight and excessive humidity, to maintain vacuum integrity and anticoagulant efficacy. Avoid extreme temperatures.
-
Q: What is the shelf life of your K2EDTA tubes?
A: Our EDTAK2 tubes typically have a shelf life of 18-24 months from the date of manufacture when stored under recommended conditions.
-
Q: Are your tubes compatible with automated hematology analyzers?
A: Yes, our EDTAK2 tubes are designed to be fully compatible with major automated hematology analyzers and laboratory automation systems due to their standardized dimensions and consistent performance characteristics.
Logistics, Warranty, and After-Sales Support
Our commitment to our B2B partners extends beyond product quality to encompass reliable logistics, comprehensive warranty, and dedicated after-sales support.
Lead Time and Fulfillment:
We maintain robust inventory levels and efficient production schedules to ensure timely fulfillment of orders. Standard lead times for regular orders range from 2-4 weeks, depending on order volume and customization requirements. For urgent requests or large-scale project rollouts, we offer expedited production and shipping options. Our global distribution network ensures reliable delivery to various international locations, adhering to cold chain requirements where applicable.
Warranty Commitments:
All EDTAK2 tubes come with a comprehensive warranty against manufacturing defects and performance issues. We guarantee that our products will meet the specified technical parameters and regulatory standards for their stated shelf life, provided they are stored and used according to instructions. Any product failing to meet these standards will be promptly replaced or credited, ensuring peace of mind for our clients.
Customer Support and Technical Assistance:
Our dedicated customer support team and technical specialists are available to provide assistance with product selection, application guidance, troubleshooting, and custom solution development. We offer multi-channel support including direct phone lines, email, and dedicated account managers to ensure swift and effective resolution of any inquiries or challenges. Regular training and updates are provided to our support staff to maintain their expertise in the latest diagnostic trends and product applications.
Conclusion
The importance of the k edta tube in modern clinical diagnostics cannot be overstated. As a critical component in ensuring accurate and reliable hematological and molecular test results, its quality directly impacts patient care and research integrity. Our EDTAK2 tubes are engineered with precision, manufactured to the highest international standards, and backed by comprehensive support services, offering an unparalleled solution for B2B partners seeking superior blood collection technology. By choosing our advanced K2EDTA tubes, laboratories and healthcare providers can enhance their diagnostic capabilities, streamline workflows, and ultimately contribute to better health outcomes.
References
- Clinical and Laboratory Standards Institute. (2017). Procedures for the Collection of Diagnostic Blood Specimens by Venipuncture; Approved Standard—Sixth Edition. CLSI document H3-A6. Wayne, PA: Clinical and Laboratory Standards Institute.
- International Organization for Standardization. (2015). ISO 6710: Single-use container111s for human venous blood specimen collection. Geneva, Switzerland: ISO.
- International Organization for Standardization. (2017). ISO 11137-1: Sterilization of health care products - Radiation - Part 1: Requirements for development, validation and routine control of a sterilization process for medical devices. Geneva, Switzerland: ISO.
- U.S. Food and Drug Administration. Medical Devices. https://www.fda.gov/medical-devices
- International Organization for Standardization. (2016). ISO 13485: Medical devices - Quality management systems - Requirements for regulatory purposes. Geneva, Switzerland: ISO.
-
Deluxe Electric Foot Warmer With Zipper-Shijiazhuang BDT Heating Pad Co.,Ltd.|Fast Heating,Customizable Design
NewsSep.02,2025 -
Deluxe Electric Foot Warmer- Shijiazhuang BDT Heating Pad Co.,Ltd.|Fast Heating,Zipper Design
NewsSep.02,2025 -
Deluxe Electric Foot Warmer- Shijiazhuang BDT Heating Pad Co., Ltd.|Fast Heating, Custom Fit
NewsSep.02,2025 -
Deluxe Electric Foot Warmer With Zipper - Shijiazhuang BDT Heating Pad Co., Ltd.
NewsSep.02,2025 -
Fast Heating Technology Deluxe Electric Foot Warmer - Shijiazhuang BDT Heating Pad Co., Ltd.
NewsSep.02,2025 -
Deluxe Electric Foot Warmer-Fast Heating Technology|Shijiazhuang BDT Heating Pad Co., Ltd.
NewsSep.02,2025














