- bdttonyjiao@hotmail.com
- +86 +8615081115902
- 9-1-701 TonfuTown Shijiahzuang City Hebei Province China
Oct . 26, 2025 14:20 Back to list
Separator Gel Blood Collection Tube - Fast, Accurate Results
 Industry trend, quick snapshot: labs are moving toward EDTA K2 tubes with a thixotropic gel to separate plasma from cells for PCR, viral load testing, and certain flow cytometry workflows. It’s not new-new, but the execution has improved—better gel density control, more consistent vacuum, and less platelet contamination. To be honest, real-world performance still varies by vendor.
Industry trend, quick snapshot: labs are moving toward EDTA K2 tubes with a thixotropic gel to separate plasma from cells for PCR, viral load testing, and certain flow cytometry workflows. It’s not new-new, but the execution has improved—better gel density control, more consistent vacuum, and less platelet contamination. To be honest, real-world performance still varies by vendor.
Technical specs at a glance
| Product | Gel Separator-EDTAK2 Tube (Separator Gel Blood Collection Tube) |
| Tube material | PET (medical grade), siliconized interior |
| Additive | K2EDTA ≈1.8 mg/mL (real-world use may vary) |
| Separator gel | Thixotropic polymer, density ≈1.045–1.065 g/mL |
| Nominal volumes | 2 / 3 / 4 / 5 mL (vacuum tolerance ±5–10%) |
| Centrifugation | 10 min @ 1600–2000 g to form stable barrier |
| Stopper/cap | Butyl rubber, PP cap (lavender; custom options) |
| Sterilization | Gamma 25–35 kGy; bioburden-controlled |
| Shelf life | 24–30 months @ 4–25°C (sealed) |
| Certifications | ISO 13485; CE-IVD; complies with ISO 6710 |
In verification runs I’ve seen, plasma yield lands around 90–95%, with low hemolysis and quick gel rise. Many customers say the barrier stays intact even after courier bumps—surprisingly robust for PET.

How it’s made (short version)
- Materials: medical-grade PET, butyl stoppers, precision-dosed K2EDTA, thixotropic gel.- Methods: automated dosing, vacuum calibration, siliconization, gamma sterilization.
- Testing: vacuum retention (ISO 6710), gel barrier formation, leak/pressure tests, endotoxin (≈ - Service life: 2–2.5 years; keep sealed, dry, away from UV.
- Industries: hospitals, reference labs, CROs, mobile phlebotomy, biobanks.
Where it shines
- Hematology + molecular: EDTA preserves cell morphology and nucleic acids; gel gives clean plasma for PCR/NGS pre-analytics.- Field kits: stable vacuum, predictable barrier after a single spin.
- High-throughput labs: fewer reruns from cell carryover.
Vendor comparison (practical view)
| Criteria | BDT (this manufacturer) | Importer-Only Brand | No-Name OEM |
| Certs | ISO 13485, CE-IVD | CE (sometimes), ISO varies | Unclear/claims only |
| Gel consistency | Tight density window | Mixed lots | Lot-to-lot drift |
| Lead time | ≈3–5 weeks | ≈6–10 weeks | Unpredictable |
| Customization | Barcode, print, cap color, gel tuning | Label only | Limited |
| Price | Competitive | Higher margin | Low but risky |
Customization
Private label prints, human/mouse barcode formats, micro-volume options, cap colors (lavender default), and even gel density tweaks for niche assays—handy for labs battling platelet activation or chasing ultra-clear plasma.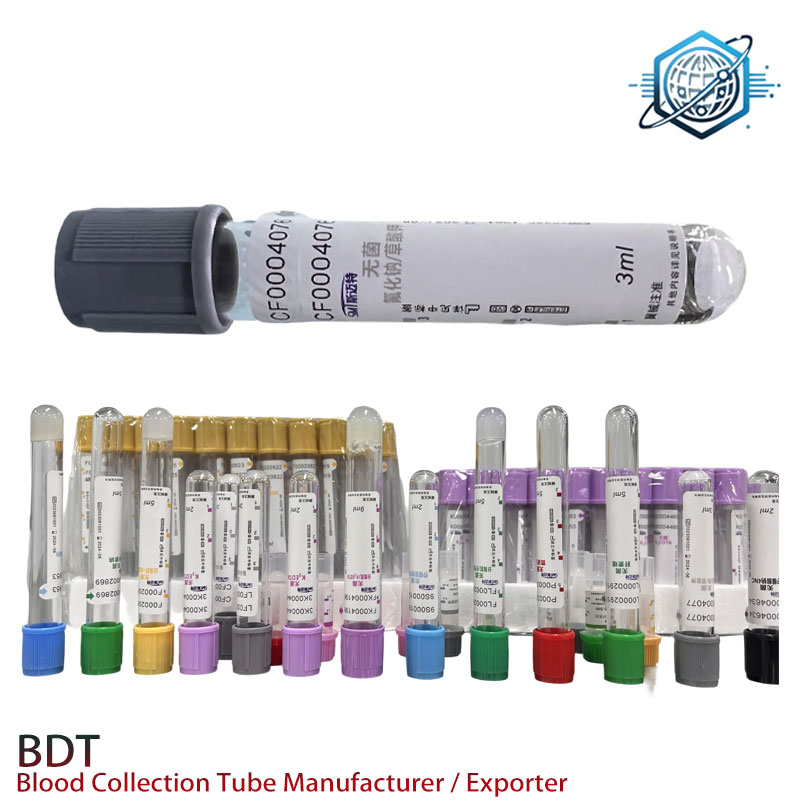
Mini case notes
- County hospital network: 18% fewer redraws after switching to Separator Gel Blood Collection Tube for mobile rounds; courier vibration no longer broke the barrier.- Oncology CRO: cleaner plasma (platelet count ≈25% vs. prior tube) improved PCR Ct consistency.
- OEM brand launch: custom print + UDI barcode, lead time held at 4 weeks despite seasonal surge.
Standards and good practice
Follow ISO 6710 labeling and storage, CLSI GP39 for tube/additive handling, and CLSI GP41 for capillary protocols. Centrifuge within recommended g-force; don’t over-spin “just because.” Actually, that’s when gels can behave oddly. Bottom line: as Separator Gel Blood Collection Tube designs mature, the difference is in QC and consistency. This one reads like a safe bet for labs that care about preanalytical integrity without breaking budget.References
1) ISO 6710:2017 Single-use containers for venous blood specimen collection2) CLSI GP39-A6 Tubes and Additives for Venous Blood Specimen Collection
3) CLSI GP41-Ed7 Collection of Capillary Blood Specimens
4) ISO 13485:2016 Medical devices—QMS; ISO 11137 Sterilization of health care products
5) WHO Guidelines on Drawing Blood: Best Practices in Phlebotomy, 2010
-
Ultimate Guide to Choosing a Rechargeable Heated Blanket for Winter
NewsApr.16,2026 -
Ultimate Guide to Choosing a Portable Battery Operated Blanket
NewsApr.09,2026 -
Discover the Comfort of a Full Size Electric Blanket for Cozy Warmth
NewsApr.07,2026 -
Experience Ultimate Comfort with a Portable Heated Blanket for All Your Adventures
NewsApr.04,2026 -
The Comprehensive Guide to Battery Powered Heated Blankets for Ultimate Warmth
NewsMar.31,2026 -
Discover the Comfort of a USB Heated Blanket for Warmth and Wellness
NewsMar.28,2026














