- bdttonyjiao@hotmail.com
- +86 +8615081115902
- 9-1-701 TonfuTown Shijiahzuang City Hebei Province China
Sep . 11, 2025 10:00 Back to list
Sodium Heparin Blood Collection Tubes - Reliable Plasma Analysis
Industry Trends and Market Dynamics
The global diagnostics market is experiencing robust growth, driven by an aging population, increasing prevalence of chronic diseases, and advancements in analytical technologies. Central to this evolution are high-quality blood collection systems. Among these, sodium heparin blood collection tubes play a critical role, particularly in clinical chemistry and blood gas analysis. The demand for precise, reliable, and contamination-free samples necessitates continuous innovation in tube manufacturing and anticoagulant quality.
Current trends indicate a strong shift towards automation and standardization in laboratories, demanding tubes that are compatible with advanced analytical platforms and reduce pre-analytical errors. Furthermore, the focus on patient safety and diagnostic accuracy has led to stricter regulatory requirements and a preference for certified, high-performance products. The market sees a growing emphasis on tubes designed for specific applications, such as the widely used green sodium heparin tube, optimized for tests requiring plasma separation without affecting enzyme activity.

Emerging markets, particularly in Asia-Pacific and Latin America, are also contributing significantly to market expansion, driven by improving healthcare infrastructure and increased access to diagnostic services. This global demand underscores the necessity for manufacturers to provide cost-effective, high-quality, and customizable solutions that meet diverse clinical needs, making the selection of the right vendor a strategic decision for healthcare providers and diagnostic centers worldwide.
Manufacturing Process Flow of Sodium Heparin Blood Collection Tubes
The production of sodium heparin blood collection tubes involves a highly controlled, multi-stage process to ensure product integrity, sterility, and optimal performance. This rigorous process is crucial for producing tubes that meet the stringent demands of modern diagnostics and maintain the quality of blood samples.
Detailed Process Steps:
1. Tube Molding/Casting
Utilizing medical-grade Polyethylene Terephthalate (PET) or Polypropylene (PP) resins, tubes are precision-molded through injection molding. This ensures consistent dimensions, wall thickness, and optical clarity. Target industries include medical device manufacturing for diagnostics.
Materials: Medical-grade PET/PP
2. Additive Coating
A precise amount of lyophilized sodium heparin is sprayed onto the inner wall of the tube. This process requires controlled environment conditions to ensure even distribution, which is critical for preventing micro-clots and ensuring accurate assay results.
Additive: Sodium Heparin
3. Drying and Capping
After coating, tubes are dried in a controlled environment to ensure complete evaporation of solvents and stable adherence of the additive. Subsequently, sterile rubber stoppers and plastic caps (e.g., green for heparin tubes) are automatically assembled under aseptic conditions.
4. Sterilization
Tubes undergo terminal sterilization, typically via irradiation (e.g., Gamma irradiation or E-beam), to achieve a Sterility Assurance Level (SAL) of 10-6. This ensures the tubes are free from viable microorganisms, meeting ISO 11137 standards.
Testing Standards: ISO 11137
5. Quality Control & Packaging
Each batch undergoes rigorous quality control, including vacuum retention tests, heparin concentration verification, and visual inspection. Certified products are then packaged in sterile, protective materials, adhering to ISO 13485 and CE standards. Typical service life is 12-24 months.
Testing Standards: ISO 13485, CE; Service Life: 12-24 months
The stringent adherence to these manufacturing protocols ensures that our sodium heparin blood collection tubes provide superior performance, critical for accurate diagnostic results across target industries such as clinical diagnostics, pharmaceutical research, and public health.
Technical Specifications and Parameters
Understanding the technical specifications of sodium heparin blood collection tubes is essential for laboratories to select the appropriate product for their analytical needs. Heparin acts by activating Antithrombin III, which inhibits various coagulation factors, particularly Thrombin and Factor Xa, thereby preventing blood clotting without significantly altering cellular morphology or interfering with most enzymatic assays.
These parameters ensure the tubes' suitability for a broad range of laboratory tests, from routine clinical chemistry to specialized analyses requiring meticulous sample preservation. The choice of the correct tube, often identifiable by its green sodium heparin tube cap, is paramount for diagnostic accuracy.
Application Scenarios
Sodium heparin blood collection tubes are indispensable in a multitude of diagnostic and research settings due to their ability to provide plasma samples suitable for a broad spectrum of tests.
- ✓ Clinical Chemistry: Heparin is the preferred anticoagulant for most clinical chemistry tests, including electrolyte panels (Na+, K+, Cl-), glucose, blood urea nitrogen (BUN), creatinine, liver function tests (ALT, AST, ALP), and cardiac enzymes (CK, CK-MB, Troponin). The advantage here is the rapid availability of plasma, allowing for quicker turnaround times compared to serum, without affecting enzyme activities.
- ✓ Blood Gas Analysis (BGA): Specifically, balanced heparin (lithium heparin or sodium heparin) is used for BGA, ensuring accurate measurement of pH, pCO2, pO2, and electrolytes crucial for assessing respiratory and metabolic states. Our tubes ensure minimal pre-analytical changes to these critical parameters.
- ✓ Therapeutic Drug Monitoring (TDM): Heparinized plasma is often suitable for TDM assays, where precise measurement of drug concentrations is vital.
- ✓ Immunology and Serology: While serum is often preferred, heparinized plasma can be used for certain immunological assays, especially when a quick result is needed.
- ✓ Research Applications: Research laboratories frequently utilize heparinized blood for various studies, including cellular analysis and plasma-based biomarker discovery.
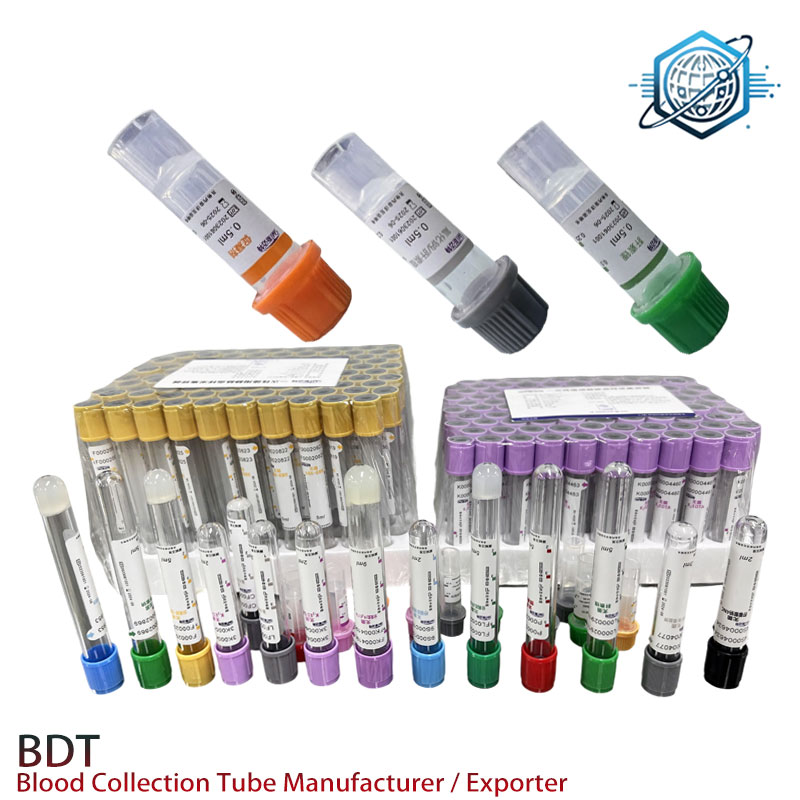
The robust design and consistent quality of our tubes ensure minimal sample hemolysis and accurate preservation of analytes, crucial for reliable diagnostic outcomes in all these scenarios. The inherent inertness of sodium heparin compared to other anticoagulants also reduces the risk of interference with a wide array of laboratory tests, making it a versatile choice for clinical laboratories.
Technical Advantages and Performance
The technical superiority of high-quality sodium heparin blood collection tubes stems from several key design and material considerations, offering significant advantages to clinical and research laboratories.
- ● Effective Anticoagulation: Sodium heparin acts swiftly and effectively, preventing coagulation by enhancing the activity of antithrombin III. This ensures a stable plasma sample suitable for immediate processing, minimizing the risk of clot formation which can interfere with automated analyzers.
- ● Minimal Interference: Unlike other anticoagulants (e.g., EDTA affecting calcium, oxalates precipitating analytes), sodium heparin generally does not interfere with enzymatic reactions or electrolyte measurements, preserving the integrity of a wide range of analytical tests. This makes it a preferred choice for comprehensive metabolic panels.
- ● Optimal Sample Quality: Our tubes are designed to prevent hemolysis during blood collection and subsequent centrifugation, crucial for obtaining high-quality plasma free from cellular contamination. The inert inner surface and precise vacuum ensure gentle blood draw.
- ● Vacuum Precision: Each tube maintains a consistent and precise vacuum, ensuring the accurate draw volume of blood. This is vital for maintaining the correct blood-to-anticoagulant ratio, a critical factor for test accuracy.
- ● Durability and Safety: Manufactured from robust, medical-grade plastics, our tubes are shatter-resistant, enhancing safety for laboratory personnel. The secure cap closure minimizes leakage and aerosol formation, adhering to global biosafety standards.
- ● Sterility and Biocompatibility: All tubes are terminally sterilized and certified to be non-pyrogenic and non-toxic, ensuring no compromise to patient samples or assay performance. This commitment to quality is underscored by ISO 13485 certification.
These advantages culminate in a product that provides unparalleled reliability and consistency, directly contributing to improved patient care through more accurate and timely diagnostic results. Our commitment to quality ensures that every sodium heparin blood collection tube performs optimally in demanding clinical environments.
Vendor Comparison and Customization
Selecting a supplier for blood collection tubes is a strategic decision for any healthcare institution or diagnostic laboratory. Factors beyond mere price, such as product quality, regulatory compliance, customization capabilities, and after-sales support, significantly impact operational efficiency and diagnostic accuracy.
Customized Solutions
Recognizing the diverse needs of modern laboratories, we offer extensive customization options for our sodium heparin blood collection tubes. This flexibility is particularly valuable for research institutions or specialized diagnostic centers with unique requirements. Our customization capabilities include:
- ✓ Volume & Size Variations: Beyond standard sizes (e.g., 2mL, 4mL, 5mL), we can produce tubes in specific draw volumes and dimensions to integrate seamlessly with unique automated systems or pediatric applications.
- ✓ Additive Concentrations: Tailored heparin concentrations to meet specific assay requirements or accommodate different patient populations.
- ✓ Special Coatings: Development of specific inner tube coatings for sensitive analytes to minimize adsorption or interaction.
- ✓ Labeling and Branding: Custom labels, barcoding, and cap colors (e.g., a sodium heparin royal blue top tube for specific research protocols requiring metal-free conditions if heparin is used as the anticoagulant) to align with institutional branding or internal laboratory protocols.
- ✓ Packaging Configurations: Customized packaging to optimize storage, handling, or compatibility with automated tube sorters.
Our dedicated team works closely with clients to understand their unique needs, providing expert consultation and rapid prototyping to deliver precisely the product required, without compromising quality or regulatory compliance.
Application Case Studies
Case Study 1: Large Hospital Network Streamlines Emergency Diagnostics
A major hospital network, comprising three urban hospitals and several outpatient clinics, faced challenges with extended turnaround times for critical emergency department (ED) lab tests. The primary issue was pre-analytical delays stemming from inconsistent blood collection tubes, leading to re-draws and sample rejections, particularly for STAT chemistry panels.
Solution: The network standardized its blood collection protocol across all facilities using our sodium heparin blood collection tubes. Our tubes were chosen for their consistent vacuum, superior heparin coating, and robust design. We provided on-site training for phlebotomy staff and integrated our tube specifications with their automated analyzers.
Results: Within six months, the hospital network reported a 15% reduction in pre-analytical errors for chemistry tests. Turnaround times for critical ED results improved by an average of 20 minutes, directly contributing to faster patient diagnoses and treatment initiation. Staff feedback highlighted the ease of use and reliability of the tubes, significantly boosting confidence in sample integrity. This improved efficiency resulted in an estimated annual saving of over $200,000 in operational costs.
Case Study 2: Research Institute Enhances Biomarker Discovery
A leading oncology research institute engaged in long-term plasma biomarker discovery studies required blood collection tubes that ensured absolute sample stability and minimal interference with sensitive proteomic assays. Their existing supplier's tubes occasionally showed trace contaminants that confounded their mass spectrometry results.
Solution: We provided customized sodium heparin blood collection tubes tailored to their stringent requirements. This involved higher-purity materials, a meticulously controlled coating process for the sodium heparin, and specialized packaging to prevent any external contamination. We also collaborated to validate the tubes' performance specifically for their proteomic workflows.
Results: The institute observed a dramatic reduction in background noise and contaminants in their mass spectrometry data, leading to a 30% increase in the detection of low-abundance biomarkers. The enhanced sample integrity significantly improved the reproducibility and reliability of their research findings, accelerating their biomarker discovery pipeline and ultimately contributing to groundbreaking publications in high-impact journals. The researchers specifically praised the consistency and purity achieved with our tailored solutions.
Frequently Asked Questions (FAQ)
Q1: What is sodium heparin and why is it used in blood collection tubes?
Sodium heparin is an anticoagulant that prevents blood clotting by enhancing the activity of antithrombin III, which in turn inhibits thrombin and factor Xa. It is used in blood collection tubes to obtain plasma samples for a wide range of clinical chemistry, blood gas, and other diagnostic tests, as it minimally interferes with most analytical assays.
Q2: What tests are compatible with sodium heparin blood collection tubes?
These tubes are primarily used for general chemistry panels, electrolyte tests, liver function tests, renal function tests, blood gas analysis, therapeutic drug monitoring, and some immunology assays. They are generally not suitable for coagulation studies or complete blood counts (CBC).
Q3: What is the shelf life of your sodium heparin blood collection tubes?
Our sodium heparin tubes typically have a shelf life of 18-24 months from the date of manufacture, provided they are stored according to recommended conditions (dry, cool place, away from direct sunlight).
Q4: Are your tubes compatible with automated blood collection and analysis systems?
Yes, our tubes are designed to meet international standards for dimensions and materials, ensuring broad compatibility with most automated blood collection, centrifugation, and analytical platforms used in modern laboratories.
Q5: How can I request a customized solution for my laboratory?
Please contact our sales or technical support team via the provided contact information. We will assign a specialist to discuss your specific requirements, including tube volume, additive concentration, labeling, or special packaging, and guide you through our customization process.
Lead Time, Warranty, and Customer Support
Lead Time and Fulfillment
We maintain efficient production and inventory management systems to ensure timely delivery. For standard orders of sodium heparin blood collection tubes, our typical lead time is 2-4 weeks from order confirmation, depending on volume and destination. Expedited shipping options are available upon request for urgent requirements. For customized solutions, lead times will be determined on a case-by-case basis after detailed discussion of specifications and production schedules. We partner with reliable logistics providers to ensure secure and efficient global delivery.
Warranty Commitments
All our products, including sodium heparin blood collection tubes, are backed by a comprehensive warranty period of 12 months from the date of shipment or the indicated shelf life, whichever comes first. This warranty covers manufacturing defects and ensures that products perform according to the specified technical parameters. We commit to replacing any defective products or issuing appropriate credits, subject to our standard terms and conditions. Our commitment reflects our confidence in the quality and reliability of our manufacturing processes.
Dedicated Customer Support
Our dedication to customer satisfaction extends beyond product delivery. We offer robust after-sales support designed to assist our B2B clients with any technical queries, operational guidance, or troubleshooting. Our expert technical support team is available via phone and email during business hours, and for critical issues, 24/7 emergency support can be arranged for key accounts. We provide:
- • Technical consultations on product application and compatibility.
- • Assistance with quality control and troubleshooting.
- • Documentation support for regulatory compliance.
- • Access to our online resource portal for product information and updates.
Our goal is to build long-term partnerships based on trust and mutual success, ensuring that our products and services consistently exceed your expectations.
Authoritative References
- Clinical and Laboratory Standards Institute (CLSI). GP41-A6. Collection of Diagnostic Venous Blood Specimens. 2017.
- International Organization for Standardization (ISO). ISO 6710:2017, Single-use container111s for human venous blood specimen collection. 2017.
- International Organization for Standardization (ISO). ISO 13485:2016, Medical devices — Quality management systems — Requirements for regulatory purposes. 2016.
- International Organization for Standardization (ISO). ISO 11137-1:2006, Sterilization of health care products — Radiation — Part 1: Requirements for development, validation and routine control of a sterilization process for medical devices. 2006.
- United States Pharmacopeia (USP). Heparin Sodium Monograph.
- European Pharmacopoeia (EP). Heparin Sodium Monograph.
-
Ultimate Guide to Choosing a Rechargeable Heated Blanket for Winter
NewsApr.16,2026 -
Ultimate Guide to Choosing a Portable Battery Operated Blanket
NewsApr.09,2026 -
Discover the Comfort of a Full Size Electric Blanket for Cozy Warmth
NewsApr.07,2026 -
Experience Ultimate Comfort with a Portable Heated Blanket for All Your Adventures
NewsApr.04,2026 -
The Comprehensive Guide to Battery Powered Heated Blankets for Ultimate Warmth
NewsMar.31,2026 -
Discover the Comfort of a USB Heated Blanket for Warmth and Wellness
NewsMar.28,2026














