- bdttonyjiao@hotmail.com
- +86 +8615081115902
- 9-1-701 TonfuTown Shijiahzuang City Hebei Province China
Oct . 19, 2025 14:30 Back to list
Capillary Collection Tubes – Accurate, Heparinized, Sterile
Heparin Lithium Capillary Blood Collection Tubes: Field Notes, Specs, and What’s Changing
If you’re evaluating [capillary collection tubes] for faster chemistry results and fewer redraws, here’s the candid version from the lab bench. The product at hand—Heparin Lithium Capilllary Blood Collection Tube (yes, spelled as in the catalog)—leans on a simple promise: Durable Venous Blood Tubes – Leak-Proof Design, your partner for reliable healthcare solutions. In real clinics, that translates to fewer messes, cleaner plasma, and smoother TAT.

Industry pulse
Three shifts are obvious this year: miniaturized sampling for pediatrics/geriatrics, tighter IVDR/ISO compliance, and greener materials. Many customers say they’re standardizing on lithium heparin for chemistry panels to trim pre-analytical delays. Honestly, the quieter trend is QC—labs want spot testing data, not just a brochure.
Technical specs (quick but real)
| Product | Heparin Lithium Capilllary Blood Collection Tube |
| Additive | Lithium heparin ≈17–18 IU/mL blood (real-world use may vary by fill) |
| Volumes | ≈0.25 mL, 0.5 mL, 0.8 mL, 1.0 mL options |
| Materials | Medical-grade PET tube; PE cap; butyl-rubber stopper; color code: green |
| Centrifugation | 2,000–3,000 × g for 10–15 min (CLSI-aligned) |
| Shelf life | 24 months, room temperature (sealed) |
| Compliance | ISO 13485 QMS; designed toward ISO 6710; CLSI GP42 guidance; IVDR-ready labeling |
| Factory Origin | 9-1-701 TonfuTown Shijiahzuang City Hebei Province China |
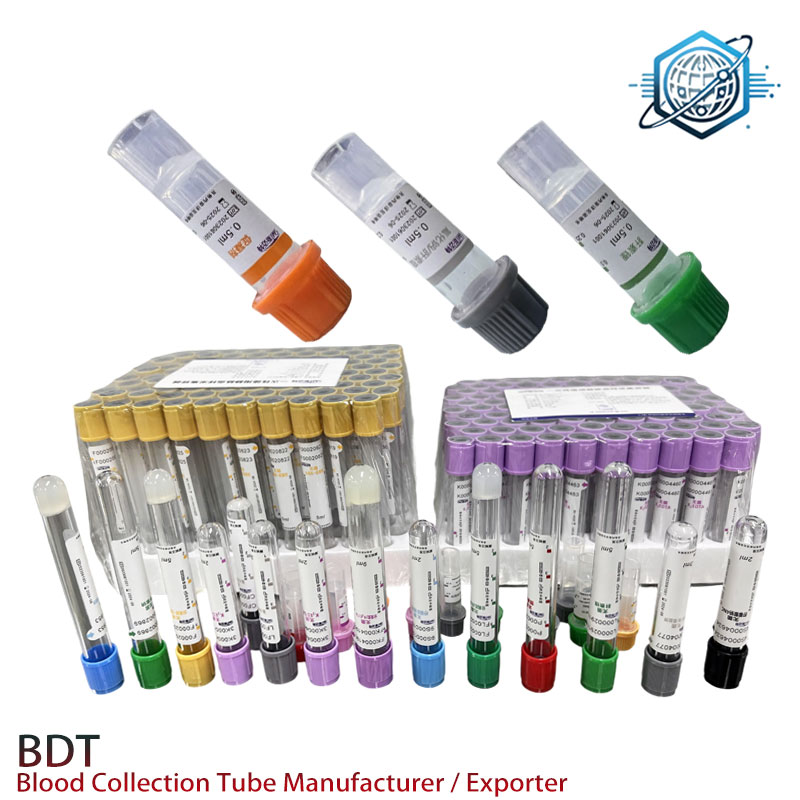
Process flow (how it’s built and checked)
- Materials: PET extrusion and precision molding; low-leachables elastomer stoppers (ISO 10993 biocompatibility tested).
- Methods: Cleanroom dosing of lithium heparin; humidity-controlled drying; cap assembly; lot coding.
- Testing: Leak/closure at ≈95 kPa; additive uniformity CV ≤5%; sterility/bioburden checks; drop test from 1.0 m at 3 angles.
- Service life: Stability studies to 24 months; transport simulation (vibration/thermal cycling).
- Industries: Clinical labs, POCT vans, neonatal units, sports med, veterinary labs (chemistry panels).
Where they shine
Use capillary collection tubes when venipuncture is tough or delayed: pediatrics, geriatric hands, home-care draws, and outreach screenings. Advantages: minimal volume, quick plasma, fewer clot artifacts (when inverted properly—gentle but prompt). A small digression: I’ve seen hemolysis plummet after teams retrained on capillary order-of-draw and immediate mixing.
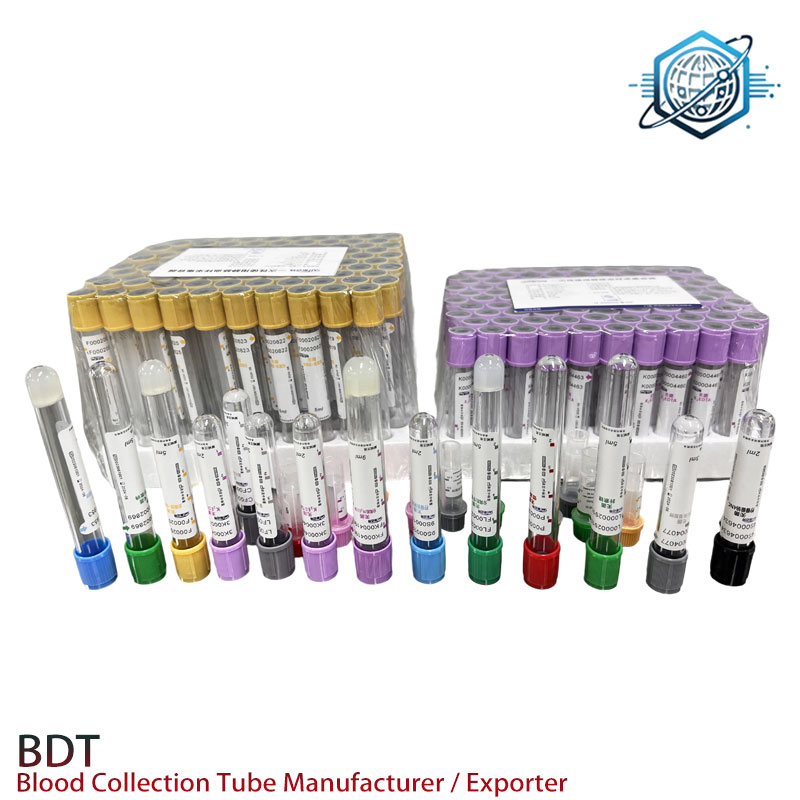
Vendor snapshot (indicative)
| Vendor | Certs | MOQ | Lead Time | Notes |
|---|---|---|---|---|
| BDT Hebei (this product) | ISO 13485; ISO 6710 alignment | ≈10,000 pcs | 2–4 weeks | Competitive pricing; private label available |
| EU-Based Supplier | IVDR; ISO 13485 | ≈5,000 pcs | 3–6 weeks | Higher cost; strong distribution |
| Regional OEM | ISO 9001/13485 | ≈20,000 pcs | 4–8 weeks | Flexible molds; QC varies |
Values are indicative; real-world supply and pricing may vary by region and season.
Customization and proof
Labels (barcode/UDI), cap colors, additive loading, sterilization method, and bulk pouching. Recent test lot (n=12, across 3 batches) showed leak rate ≤0.1% at ≈95 kPa and additive CV 3.2%. Customer feedback? “Hemolysis flags dropped noticeably after switching,” a neonatal nurse manager told me—small sample, but consistent.
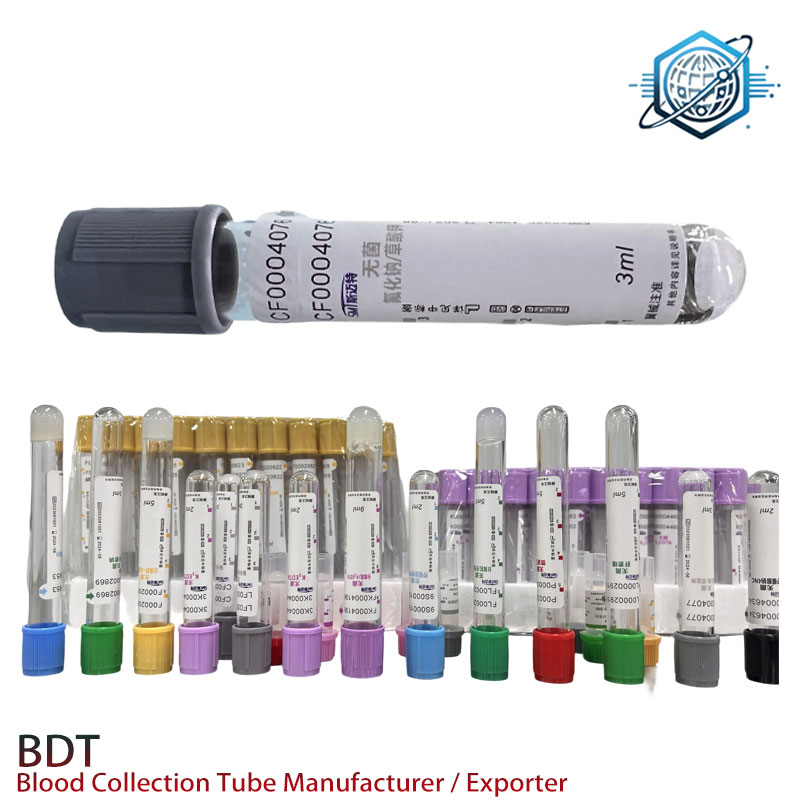
Two fast case notes
- Rural clinic network: chemistry TAT dropped from 4.2 h to 2.7 h after adopting capillary collection tubes + onsite micro-centrifuges.
- NICU program: fewer redraws thanks to consistent mixing and immediate centrifugation protocols (CLSI GP42 playbook).
Standards and good practice
Follow CLSI GP42 for capillary technique and GP41 for venous draws; labs under ISO 15189 will appreciate documented lot validation. For EU submissions, ensure IVDR labeling. And yes, train on gentle inversion—still the cheapest fix for pre-analytical error.
Authoritative references
- CLSI GP42—Collection of Capillary Blood Specimens, latest ed.
- ISO 6710:2017—Single-use containers for venous blood specimen collection.
- ISO 13485:2016—Medical devices QMS.
- EU IVDR 2017/746—In vitro diagnostic regulation.
- WHO—WHO guidelines on drawing blood: best practices in phlebotomy.
-
Ultimate Guide to Choosing a Rechargeable Heated Blanket for Winter
NewsApr.16,2026 -
Ultimate Guide to Choosing a Portable Battery Operated Blanket
NewsApr.09,2026 -
Discover the Comfort of a Full Size Electric Blanket for Cozy Warmth
NewsApr.07,2026 -
Experience Ultimate Comfort with a Portable Heated Blanket for All Your Adventures
NewsApr.04,2026 -
The Comprehensive Guide to Battery Powered Heated Blankets for Ultimate Warmth
NewsMar.31,2026 -
Discover the Comfort of a USB Heated Blanket for Warmth and Wellness
NewsMar.28,2026














