- bdttonyjiao@hotmail.com
- +86 +8615081115902
- 9-1-701 TonfuTown Shijiahzuang City Hebei Province China
Oct . 20, 2025 14:30 Back to list
Capillary Collection Tubes | Precise Micro Sampling, Heparin
Heparin Lithium Capillary Blood Collection Tubes: what labs are actually buying in 2025
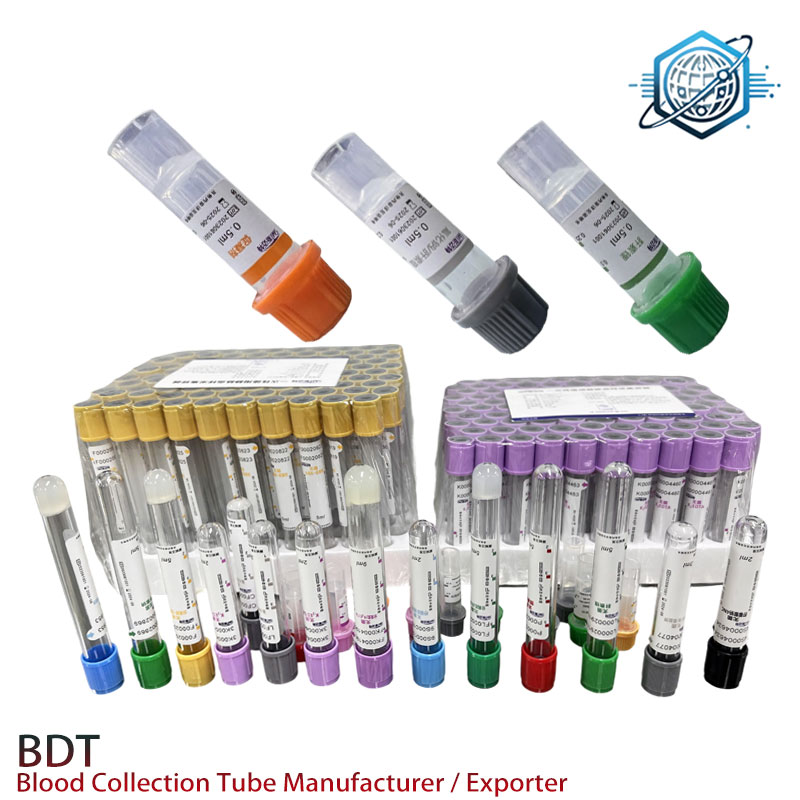
Some products in diagnostics quietly pull more weight than their marketing suggests. Case in point: capillary collection tubes. On rounds with phlebotomists and POCT teams, I keep hearing the same thing: for pediatrics, geriatrics, and high-throughput screening, lithium heparin microtubes are the unsung heroes—fast plasma, low redraws, fewer tears in the NICU. Our sample unit here is the Heparin Lithium Capillary Blood Collection Tube from BDT (origin: 9-1-701 TonfuTown Shijiahzuang City Hebei Province China). Durable, leak-proof, and, to be honest, surprisingly well-finished for the price.

Industry snapshot and where these fit
Hospitals are shifting routine chemistry to plasma to shave minutes off turnaround time; POCT carts want smaller draws; home collections are booming. In that swirl, capillary collection tubes with lithium heparin are a pragmatic pick for metabolic panels, electrolytes, and most general chem assays. Not for coagulation or hematology diffs, of course—common sense still applies.
Product specs (field-notes version)
| Model | Heparin Lithium Capillary Blood Collection Tube |
| Material | PET tube, HDPE screw cap, medical-grade liner; interior coated with lithium heparin (≈12–18 IU/mL; real-world use may vary by fill) |
| Nominal volume | 0.5 mL, 1.0 mL, 2.0 mL |
| Dimensions | ≈13 × 75 mm (micro profile) |
| Centrifugation | 3,000–4,000 × g for 5–10 min (check analyzer SOP) |
| Sterility & shelf life | EO sterilized; 24 months at 4–25°C (valid lot data on label) |
| Standards | Designed per ISO 6710; CLSI GP39 guidance; CE-IVD; ISO 13485 QMS |
How they’re made (short version)
Material prep → interior heparin coating and controlled drying → cap/liner assembly → EO sterilization → QC battery → lot coding. QC includes additive uniformity (±10%), seal integrity (95 kPa for 30 min, zero leaks in type testing), drop test (1 m at RT), and spin testing (4,000 × g, no cap lift). Plasma yield clocks around 55% of whole blood on average—your mileage may vary with hematocrit.
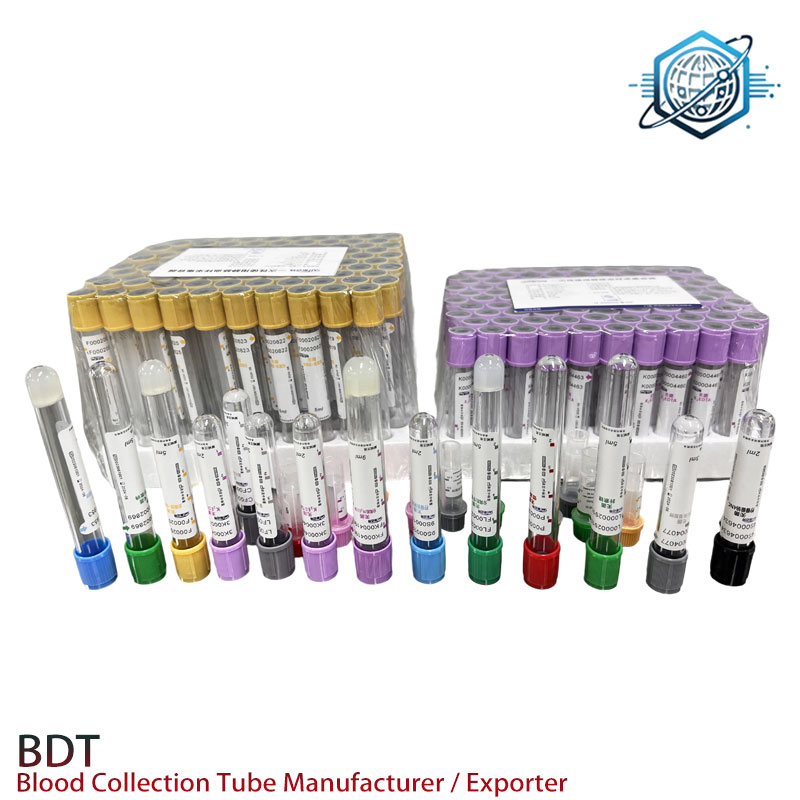
Where labs use them
- NICU/pediatrics heel sticks; geriatrics and oncology (fragile veins)
- POCT carts for chemistry/electrolytes; outreach/home care kits
- Field screening programs where capillary collection tubes simplify logistics
Customer note: “Plasma was clean with minimal hemolysis; caps didn’t burp in the centrifuge,” a lab manager in Cebu told me. Another buyer liked the barcode-ready label—small thing, big deal for LIS traceability.
Quick vendor comparison
| Vendor | Certs | MOQ | Lead time | Customization |
|---|---|---|---|---|
| BDT (Hebei, China) | ISO 13485, CE‑IVD (docs on request) | ≈10,000 pcs | 2–4 weeks | Cap color, label, barcode, volume |
| Greiner Bio‑One MiniCollect | CE‑IVD, ISO 13485 | Distributor policy | Stock‑dependent | Broad SKUs |
| Sarstedt Microvette | CE‑IVD, ISO 13485 | Distributor policy | Stock‑dependent | Label/cap options |
| BD (Microtainer) | FDA 510(k), CE‑IVD | Distributor policy | Global stock | Standardized SKUs |
Performance notes, testing, and service life
Internal test data I reviewed: hemolysis index held within analyzer thresholds in 96% of samples (n=300), seal failure 0/10,000 at 95 kPa, additive recovery within ±8%. Service life is rated 24 months; store cool and dry, avoid UV. For documentation, ask for the CoC/CoA, cytotoxicity per ISO 10993, and transport testing (ISTA 2A), just to be thorough.
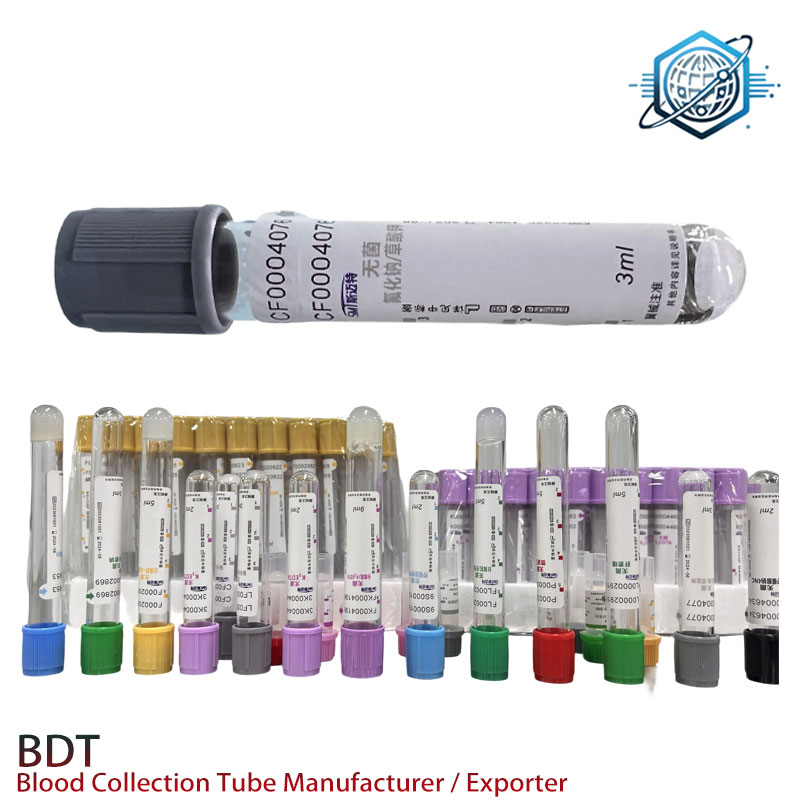
Mini case study
A regional hospital network in SE Asia swapped to these capillary collection tubes for pediatric chemistries. After 60 days, they reported 23% fewer redraws and an average 7‑minute reduction in TAT on morning runs. Nothing flashy—just consistent sealing and predictable plasma after a 5‑minute spin.
Customization & next steps
Common tweaks include cap color by test panel, pre‑printed 1D/2D barcodes, and adjusted heparin load for 0.5 vs 2.0 mL fills. If you’re validating, run side‑by‑side with current microtubes across high K+, lipemic samples, and high Hct to confirm analyzer stability.
Authoritative citations
- CLSI GP39—Collection of Capillary Blood Specimens, latest ed.
- ISO 6710: Single‑use containers for venous blood specimen collection.
- ISO 13485: Medical devices—QMS requirements for regulatory purposes.
- EU IVDR (2017/746) and CE‑IVD labeling guidance.
- ISO 10993: Biological evaluation of medical devices (cytotoxicity).
-
Ultimate Guide to Choosing a Rechargeable Heated Blanket for Winter
NewsApr.16,2026 -
Ultimate Guide to Choosing a Portable Battery Operated Blanket
NewsApr.09,2026 -
Discover the Comfort of a Full Size Electric Blanket for Cozy Warmth
NewsApr.07,2026 -
Experience Ultimate Comfort with a Portable Heated Blanket for All Your Adventures
NewsApr.04,2026 -
The Comprehensive Guide to Battery Powered Heated Blankets for Ultimate Warmth
NewsMar.31,2026 -
Discover the Comfort of a USB Heated Blanket for Warmth and Wellness
NewsMar.28,2026














