- bdttonyjiao@hotmail.com
- +86 +8615081115902
- 9-1-701 TonfuTown Shijiahzuang City Hebei Province China
Oct . 10, 2025 09:35 Back to list
Capillary Collection: Safer, Faster, Low-Volume Tubes?
Capillary Tube – Field Notes from a Lab Gear Insider
If you work in pediatrics, POCT, or mobile phlebotomy, you already know the quiet importance of capillary collection. I’ve toured factories from Hebei to Hamburg, and—honestly—what separates good micro-collection tubes from forgettable ones is a blend of materials science, dosing precision, and surprisingly, cap design. The product in focus today: Capillary Tube – Capillary Blood Collection Tube (Origin: 9-1-701 TonfuTown Shijiahzuang City Hebei Province China). Global trade show exhibitor, with on-ground support in North America and Europe. That last bit matters when your field team needs help, fast.

Industry trends (short version)
- Shift to decentralized testing and home sampling is accelerating.
- EDTA microtubes with low hemolysis rates are winning pediatric labs.
- Buyers want CE-IVD, ISO 13485, and a predictable shelf life. And yes, real-world throughput beats brochure specs.
Technical overview and specs
Materials: medical-grade PET tube, PP closure, options for K2EDTA (spray), clot activator (silica), or plain. EO sterilization. Typical shelf life: ≈24 months (around 4–25°C storage). Centrifugation tolerance up to ≈5,000 × g (real-world use may vary).
| Parameter | Typical Value | Notes |
|---|---|---|
| Fill volume | 200–500 μL | Low sample burden for neonates/geriatrics |
| Additive accuracy | ±10% | Per lot, gravimetric check |
| Hemolysis control | Low (qualitative) | Technique-dependent, see CLSI guidance |
| Seal integrity | >20 N | No leakage in tip-over test (n=500) |
| Certifications | ISO 13485, CE-IVD | Documentation on request |
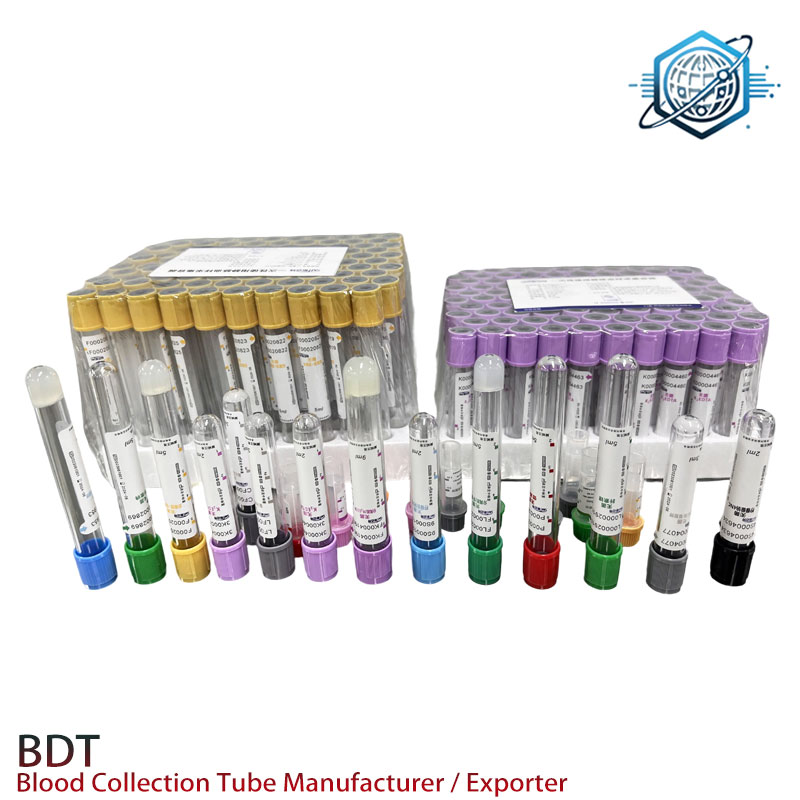
Process flow, testing standards, and service life
Manufacturing flow (simplified): raw material incoming QC → additive dosing (EDTA/silica) → drying/curing → EO sterilization → cap assembly and torque check → lot sampling (additive mass, seal, bioburden) → labeling/barcode → packaging. Standards referenced: CLSI GP42 for capillary blood procedures, ISO 6710 for specimen container111s, ISO 10993 for biocompatibility elements, and IVDR documentation. Routine type tests include drop tests, cap retention, particulate limits, and centrifuge endurance. Shelf life verification uses real-time and accelerated aging; I’ve seen lots hold well past 24 months, but the label stays conservative (as it should).
Applications and scenarios
- Pediatrics/neonatology where venipuncture is tough.
- POCT carts in urgent care.
- Home-health and outreach programs (small volume, simple handling).
- Sports medicine and wellness screening; also veterinary clinics.
Many customers say the switch to micro-collection reduced redraws, which matches what I’ve seen—when staff follows best practices for capillary collection.
Vendor comparison (quick reality check)
| Vendor | Certs | MOQ | Customization | Lead Time | Support |
|---|---|---|---|---|---|
| Hebei Manufacturer (this product) | ISO 13485, CE-IVD | ≈10,000 pcs | Cap color, barcode, private label, fill volume | 3–5 weeks | NA/EU on-ground reps |
| EU Brand A | CE-IVD, ISO 13485 | ≈5,000 pcs | Limited colors, fixed volumes | 2–4 weeks | EU-focused |
| Generic Importer | Varies | Low | Unclear | Unpredictable | Email-only |
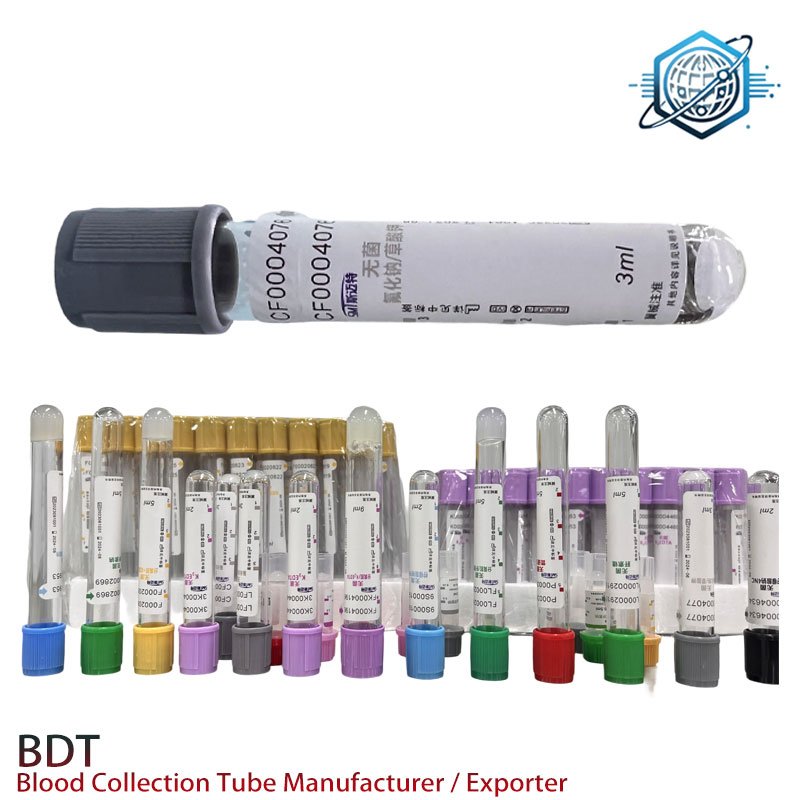
Customization and field feedback
Options I’ve seen work well: color-coded caps by analyte, pre-applied 1D/2D barcodes, bilingual labels, and EDTA mass tuned to target Hct ranges. One buyer in Ontario said, “Barcode-ready packs cut relabel errors in half.” Another from a pediatric lab in Spain noted fewer clots after switching to silica-activated microtubes for chemistry—technique and training still rule, but the hardware helps.
Case snapshots
- US pediatric lab: standardizing micro-collection workflow (per CLSI guidance) dropped redraws by ≈12% over three months.
- EU urgent care chain: private-labeled tubes with QR codes sped intake; the surprising win was faster centrifuge balancing with consistent 300 μL fills during capillary collection.
Bottom line
If your teams live in the world of small volumes and high stakes, pick tubes with verified additive accuracy, robust caps, and proper documentation. On-ground support is not a luxury—it’s uptime.
Authoritative citations
- CLSI GP42. Procedures and Devices for the Collection of Diagnostic Capillary Blood Specimens.
- ISO 6710:2017. Single-use container111s for human venous blood specimen collection (applicable principles).
- ISO 13485:2016. Medical devices—Quality management systems—Requirements for regulatory purposes.
- ISO 10993 series. Biological evaluation of medical devices (selected parts for materials).
- EU Regulation 2017/746 (IVDR) and FDA 21 CFR 820 Quality System Regulation (documentation and QMS).
-
Ultimate Guide to Choosing a Rechargeable Heated Blanket for Winter
NewsApr.16,2026 -
Ultimate Guide to Choosing a Portable Battery Operated Blanket
NewsApr.09,2026 -
Discover the Comfort of a Full Size Electric Blanket for Cozy Warmth
NewsApr.07,2026 -
Experience Ultimate Comfort with a Portable Heated Blanket for All Your Adventures
NewsApr.04,2026 -
The Comprehensive Guide to Battery Powered Heated Blankets for Ultimate Warmth
NewsMar.31,2026 -
Discover the Comfort of a USB Heated Blanket for Warmth and Wellness
NewsMar.28,2026














