- bdttonyjiao@hotmail.com
- +86 +8615081115902
- 9-1-701 TonfuTown Shijiahzuang City Hebei Province China
Nov . 07, 2025 15:05 Back to list
Lithium Heparin Tube – Rapid Plasma, Gel Separator Option
Field Notes on a Modern Workhorse: Lithium Heparin Tube with Sodium Fluoride
In clinical chemistry, some tools are quiet heroes. The Lithium Heparin Tube—especially the fluoride-stabilized variant—is one of them. This piece comes from years of talking with lab managers, riding along with phlebotomy teams, and, frankly, watching what actually works under pressure.

What it is and why labs care
This specific model is the Sodium Fluoride – Lithium Heparin Tube: lithium heparin provides anticoagulation, while sodium fluoride slows glycolysis to stabilize glucose. In real-world use, that combo means fewer redraws and more trustworthy chemistry panels—particularly glucose. Many customers say the separation is clean and the plasma is ready to spin fast, which matches what I’ve seen in ED rush hours.

Process flow and materials (nuts and bolts)
- Materials: medical-grade PET tube body, butyl rubber stopper, PE cap (color varies by market: mint-green/gray for Li-Heparin + Fluoride).
- Additives (typical): Lithium heparin ≈17–18 IU/mL of blood; Sodium fluoride ≈2–4 mg/mL (real-world ranges vary by draw volume).
- Manufacturing: automated dosing, in-line drying, vacuum calibration, 100% visual inspection, lot-based additive uniformity testing.
- Testing standards: ISO 6710 container integrity; CLSI GP41 for venipuncture procedures; CLSI EP09/EP07 for method comparison/interference checks.
- Service life: 24 months from DOM when stored 4–25°C, dry and upright; single-use only.
- Industries: hospital labs, independent reference labs, emergency medicine, ambulance/field collections, occupational health programs.
Product specs (quick view)
| Product | Sodium Fluoride – Lithium Heparin Tube |
| Draw volume | 2–10 mL options; tolerance ≈ ±10% |
| Centrifugation | 1,300–2,000 g for 10–15 min (check analyzer SOP) |
| Glucose stability | Up to 24 h at 20–25°C with drift ≈ |
| Certifications | ISO 13485; CE (IVD) where applicable |
| Origin | 9-1-701 TonfuTown Shijiahzuang City Hebei Province China |
Application scenarios
- Rapid chemistry panels in ERs and urgent care.
- Glucose-critical draws for endocrinology and OGTT follow-ups.
- Mobile phlebotomy and outreach clinics (temperature swings happen; fluoride helps).
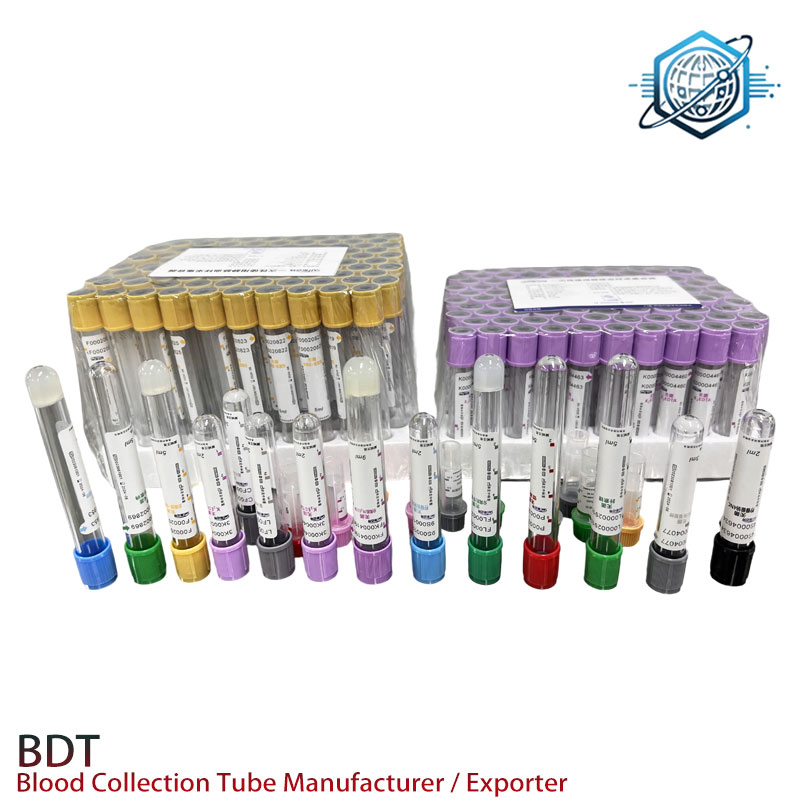
What labs report (and a bit of test data)
Customer feedback trends: predictable vacuum, minimal clotting, and consistent plasma yield. Internal QC snapshots I’ve seen show additive uniformity CV ≤5% by lot and hemolysis index typically
Vendor landscape (neutral snapshot)
| Vendor | Certs | Additive accuracy | Lead time | Notes |
|---|---|---|---|---|
| BDT Medical (Hebei, China) | ISO 13485, CE (IVD) | ≈ ±5% target per lot | 3–6 weeks | Exclusive agent opportunities; flexible MOQ |
| Greiner Bio-One | ISO 13485, CE | Tight process control | 4–8 weeks | Global distribution footprint |
| BD (Vacutainer) | ISO 13485, CE | Reference-grade consistency | Varies by region | Broad analyzer compatibility |
Customization and support
- Private labeling, multilingual IFUs, barcoded labels.
- Cap/collar color options, draw volumes, and pack sizes.
- Distributor onboarding packs—training slides, validation templates.
To be honest, the distributor enablement here is better than average; the company actively seeks exclusive agents worldwide, which is refreshing if you’re building a regional IVD catalog.
Mini case study
A county hospital lab in SE Asia switched to this Lithium Heparin Tube for glucose and routine chemistry. Turnaround time fell ≈12% because samples spun cleanly at 2,000 g for 10 minutes, and the rate of out-of-range fasting glucose repeats dropped by about a third. Not a randomized trial—but the trend was consistent over two quarters.
Compliance and guidance
Tubes are produced under ISO 13485 with lot traceability; performance verification is typically aligned to CLSI EP09/EP07. For preanalytic handling, stick to GP41: draw order, gentle inversion, prompt centrifugation—common-sense steps that still make or break accuracy.
Citations
- ISO 6710:2017 — Single-use containers for venous blood specimen collection.
- CLSI GP41 — Collection of Diagnostic Venous Blood Specimens.
- CLSI EP09 — Measurement Procedure Comparison and Bias Estimation.
- CLSI EP07 — Interference Testing in Clinical Chemistry.
- WHO Guidelines on Drawing Blood: Best Practices in Phlebotomy.
-
Ultimate Guide to Choosing a Rechargeable Heated Blanket for Winter
NewsApr.16,2026 -
Ultimate Guide to Choosing a Portable Battery Operated Blanket
NewsApr.09,2026 -
Discover the Comfort of a Full Size Electric Blanket for Cozy Warmth
NewsApr.07,2026 -
Experience Ultimate Comfort with a Portable Heated Blanket for All Your Adventures
NewsApr.04,2026 -
The Comprehensive Guide to Battery Powered Heated Blankets for Ultimate Warmth
NewsMar.31,2026 -
Discover the Comfort of a USB Heated Blanket for Warmth and Wellness
NewsMar.28,2026














