- bdttonyjiao@hotmail.com
- +86 +8615081115902
- 9-1-701 TonfuTown Shijiahzuang City Hebei Province China
Oct . 16, 2025 15:05 Back to list
Sodium EDTA Tube – Accurate Hematology, Sterile, Leak-Proof
EDTANa2 Tube: Field Notes, Specs, and What Labs Really Care About
If you’ve handled hematology samples this week (who hasn’t?), you’ve probably reached for a sodium edta tube. The model I’ve been seeing more lately is the EDTANa2 Tube from a factory in 9-1-701 TonfuTown, Shijiahzuang City, Hebei, China—quietly supplying big reference labs and, increasingly, mobile phlebotomy teams. Global partner wanted, they say. Honestly, the timing makes sense: procurement teams want stable vacuum, clean lot-to-lot EDTA, and real customization without the drama.
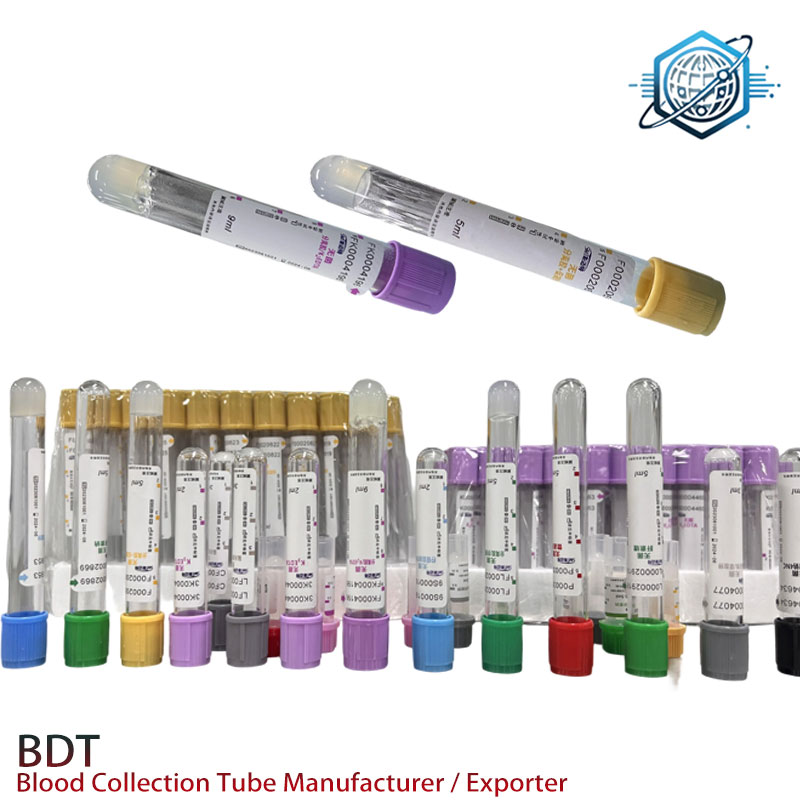
Industry snapshot (and what’s shifting)
Two big trends: tighter preanalytical control and serialization. Labs are asking for barcode-ready tubes, EO sterilization proof, and documented EDTA activity. Also, to be honest, many procurement teams are mixing premium and value vendors to hedge risk—especially when ESR and CBC volumes spike seasonally.
Product specs at a glance
| Parameter | EDTANa2 Tube (typical) |
|---|---|
| Anticoagulant | Disodium EDTA (Na2EDTA), USP/EP grade, spray-dried |
| EDTA dose | ≈1.5–2.0 mg/mL of intended fill (real-world use may vary) |
| Tube material | PET standard; glass optional |
| Draw volumes | 2, 3, 4, 5, 6, 10 mL |
| Vacuum tolerance | Fill accuracy ±10% per ISO 6710 |
| Cap/stopper | Butyl stopper; color options incl. lavender; leak-tested |
| Sterilization | EO sterilized; bioburden control per ISO 11737 |
| Shelf life | 24 months (vacuum retention validated) |
| Standards/certs | ISO 13485; CE (IVDR); compliant with ISO 6710, CLSI GP39 |
Where it’s used
Hematology (CBC/differential), blood banking screens, DNA workflows, and select trace-element protocols where potassium contamination is a concern. Note: sodium-based EDTA isn’t ideal when sodium measurement itself is the analyte of interest.

Process flow (how it’s made and verified)
- Materials: medical-grade PET/glass; pharmaceutical-grade Na2EDTA; butyl stoppers; HDPE caps.
- Methods: spray-coating EDTA; vacuum set; siliconization (as needed); cap assembly; EO sterilization.
- Testing: draw volume accuracy; EDTA content assay (complexometric titration); hemolysis index; stopper leak; endotoxin screening; sterility per ISO 11737; label adhesion/print durability.
- Service life: validated for 24 months storage; single-use only.
- Industries: hospitals, reference labs, CROs, blood banks, veterinary labs, mobile phlebotomy.
Vendor comparison (real procurement talk)
| Vendor | MOQ | Lead time | Customization | Certs | Price/1k (EXW) |
|---|---|---|---|---|---|
| Hebei EDTANa2 (Manufacturer) | ≈10k pcs | 3–5 weeks | Logo, barcode, EDTA dose, cap color | ISO 13485, CE | $25–$38 |
| Global Brand A | ≈5k pcs | 6–10 weeks | Limited | ISO 13485, CE, 510(k) | $40–$60 |
| OEM B | ≈50k pcs | 5–7 weeks | Wide, but tooling fees | ISO 13485 | $20–$30 |
Data indicative; regional logistics and resin markets can nudge prices and lead times.
Advantages and feedback
- Consistent anticoagulation: labs report clean smears and stable MCV/PLT at 2–6 hours post-draw.
- Reliable vacuum: fewer underfills on mobile rounds—many customers say that’s the quiet win.
- Customization: private label, lot-level barcoding, and yes, lavender caps that actually match your rack set.
Mini case
A mid-size Southeast Asian reference lab moved to sodium edta tube Na2 for hematology and genetics prep. Underfill rate dropped from 3.1% to 1.2% over 8 weeks; hemolysis index outliers decreased ≈18% after staff retraining and switching to PET. Not magic—just consistent consumables plus better phlebotomy technique.
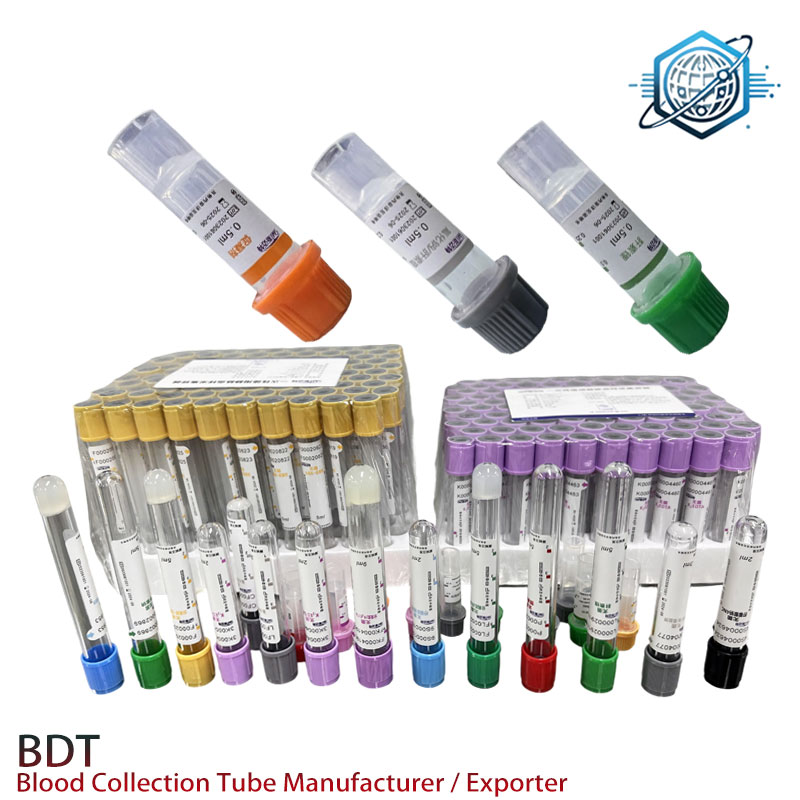
Quality and compliance checkpoints
Look for ISO 13485 certification and documented conformity with ISO 6710 and CLSI GP39. Ask for EDTA assay reports, vacuum retention curves, and EO residuals. For regulated markets, confirm CE (IVDR) and—if applicable—FDA 510(k) status. Simple rule: if a vendor hesitates on lot COAs, move on.
Final note
The sodium edta tube space isn’t flashy, but reliability pays for itself. This Hebei manufacturer is openly seeking global partners; if you need steady supply plus customization, it’s worth a pilot lot.
References
- CLSI GP39—Collection of Diagnostic Venous Blood Specimens, 6th ed.
- ISO 6710:2017/2023—Single-use containers for venous blood specimen collection.
- WHO—Guidelines on Drawing Blood: Best Practices in Phlebotomy, 2010.
- EFLM—Hemolysis management recommendations, 2020.
- FDA—Device 510(k) Premarket Notification Database (accessed regularly by procurement teams).
-
Ultimate Guide to Choosing a Rechargeable Heated Blanket for Winter
NewsApr.16,2026 -
Ultimate Guide to Choosing a Portable Battery Operated Blanket
NewsApr.09,2026 -
Discover the Comfort of a Full Size Electric Blanket for Cozy Warmth
NewsApr.07,2026 -
Experience Ultimate Comfort with a Portable Heated Blanket for All Your Adventures
NewsApr.04,2026 -
The Comprehensive Guide to Battery Powered Heated Blankets for Ultimate Warmth
NewsMar.31,2026 -
Discover the Comfort of a USB Heated Blanket for Warmth and Wellness
NewsMar.28,2026














