- bdttonyjiao@hotmail.com
- +86 +8615081115902
- 9-1-701 TonfuTown Shijiahzuang City Hebei Province China
Oct . 24, 2025 16:20 Back to list
Lithium Heparin Separator Tube – Fast Plasma, Gel Precision
Gel Separator–Lithium Heparin Tube: what labs are really asking for in 2025
If you spend time on the chemistry bench (or you’re the unlucky soul troubleshooting hemolysis spikes after a centrifuge swap), you know where the conversation is going: the Lithium Heparin Separator Tube is becoming the workhorse for rapid plasma testing. Under the hood, this “green-top with gel” blends a thixotropic polymer barrier with lithium heparin anticoagulant—meaning faster turnaround, less clotting drama, and workflows that behave well with automation lanes. From a market angle, there’s clear momentum toward PET-based tubes, cleaner gel systems, and tighter vacuum tolerances. To be honest, the quiet win is consistency.
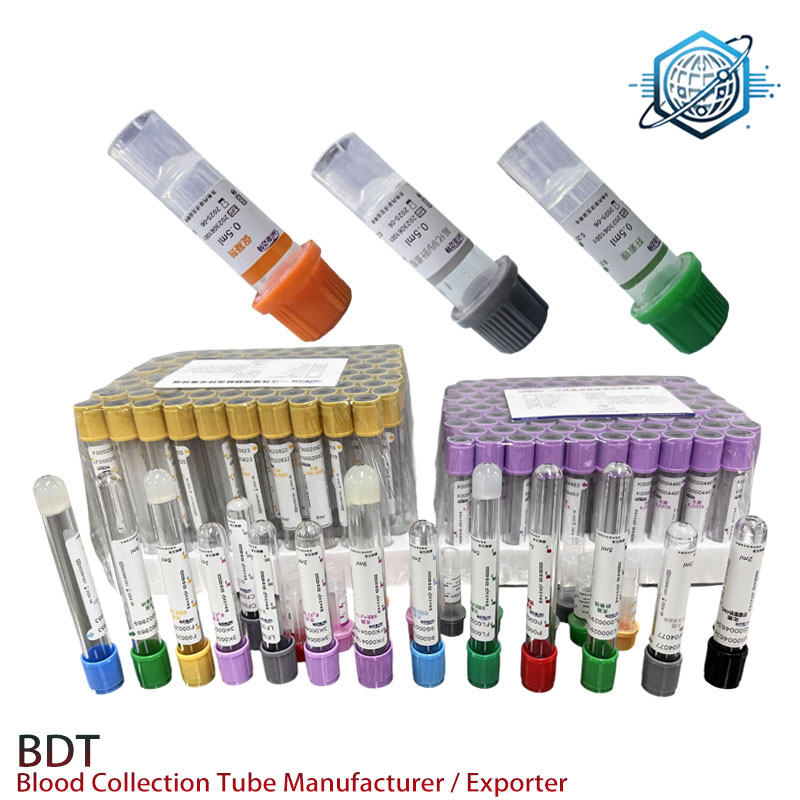
What it is and where it’s from
Product name: Gel Separator–Lithium Heparin Tube. Origin: 9-1-701 TonfuTown, Shijiahzuang City, Hebei Province, China. The supplier positions itself as a “Custom Blood Collection Tubes Supplier – Collaborate for Worldwide Market Success,” which, in practice, has meant flexible MOQs, private labeling, and surprisingly fast die-line work for barcodes. Many customers say the caps reseal cleanly—small detail, big morale boost for phlebotomy.
Typical applications
- Core chemistry panels and STAT plasma testing (ALT/AST, electrolytes, lipids; real-world use may vary).
- Automated analyzers with primary-tube sampling; compatible with standard decappers.
- Not for lithium therapeutic drug monitoring (obviously), and generally not for coag or PCR workflows.
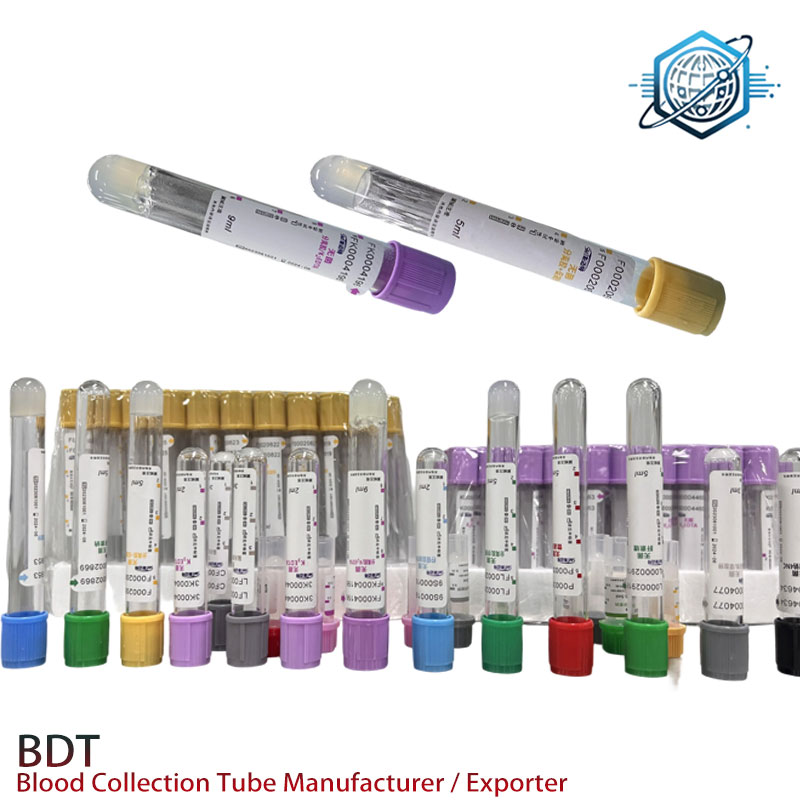
Technical specifications (field-proven, lab-friendly)
| Tube material | PET (copolyester), clear, low breakage |
| Stopper/cap | Butyl rubber with green cap; closure torque ≈ standard automation spec |
| Additive | Lithium heparin 17–20 IU/mL (spray-coated) |
| Separator gel | Thixotropic polymer, SG ≈1.045 g/mL, 0.8–1.2 g per tube |
| Sizes/draw | 13×75 mm (3–4 mL), 13×100 mm (4–5 mL) |
| Centrifugation | 1,300–2,000 g for 10 min (verify per analyzer/rotor) |
| Vacuum accuracy | ±10% of nominal at 20–25°C (ISO 6710 guidance) |
| Shelf life | 18–24 months at 4–25°C; keep dry, upright |
| Post-spin stability | Gel barrier integrity up to 48 h at 2–8°C; analyte stability depends on test menu |
| Certifications | ISO 13485 QMS; CE-IVD; biocompatibility per ISO 10993 (supplier data) |
Internal QC snapshots (n≈600 across three sites): hemolysis index typically low, with plasma yield >90% after recommended spin; barrier slumping not observed at 37°C within 8 h. Your mileage may vary—rotor geometry and g-force calibration matter more than people think.
Process flow, materials, testing
Materials include PET tubes, butyl stoppers, and a medical-grade gel. Methods: in-line additive spray, precision gel dosing, vacuum setting, and leak/vacuum verification. Testing standards reference ISO 6710 for draw accuracy, CLSI GP41 for collection, and routine pull/torque, drop, and transport vibration tests. Service life runs through expiration; barrier performance is rechecked by accelerated aging and lot validation.
Vendor landscape (quick comparison)
| Vendor | Certs | Customization | Lead time | Price tier |
|---|---|---|---|---|
| Hebei OEM (ShijiazhuANG) | ISO 13485, CE-IVD | High (color, volumes, labels, barcodes) | 3–6 weeks (≈, order size) | Value |
| Global Brand A | ISO 13485, IVDR-ready | Medium | 4–10 weeks | Premium |
| Global Brand B | ISO 13485, CE-IVD | Low–Medium | 6–12 weeks | Premium |
Customization notes
Private label kits, different green shades, 2–6 mL draw volumes, pre-attached barcode ranges, and packaging for hot/humid lanes are available. I guess the sleeper feature is vacuum tuning for high-altitude sites—saves headaches.

Case study (MENA regional hospital network)
After switching to Lithium Heparin Separator Tube lots customized for 4 mL draws and barcode A5 labels, a 9-site network reported an ≈18% reduction in recollects linked to partial draws over 90 days. TAT for core chem dropped by ~7 minutes per sample in their STAT stream. Feedback from phlebotomy: “smooth draw, fewer short-samples.” Caveat: they upgraded centrifuge inserts at the same time—so, yes, confounders.
Advantages (day-to-day reality)
- Fast plasma availability without clot wait times.
- Stable gel barrier that survives routine transport vibration.
- Lower breakage vs. glass; better for high-throughput analyzers.
Standards and good practice still apply: follow CLSI collection/processing, verify centrifuge g-force, and avoid Lithium Heparin Separator Tube for lithium assays or tests where heparin interferes.
Authoritative citations
- CLSI GP41 (Collection of Diagnostic Venous Blood Specimens), latest edition.
- ISO 6710:2017 – Single-use containers for venous blood specimen collection.
- CLSI GP44 – Procedures for Handling and Processing of Blood Specimens.
- ISO 13485:2016 – Medical devices – Quality management systems.
- Regulation (EU) 2017/746 (IVDR) – In vitro diagnostic medical devices.
-
Ultimate Guide to Choosing a Rechargeable Heated Blanket for Winter
NewsApr.16,2026 -
Ultimate Guide to Choosing a Portable Battery Operated Blanket
NewsApr.09,2026 -
Discover the Comfort of a Full Size Electric Blanket for Cozy Warmth
NewsApr.07,2026 -
Experience Ultimate Comfort with a Portable Heated Blanket for All Your Adventures
NewsApr.04,2026 -
The Comprehensive Guide to Battery Powered Heated Blankets for Ultimate Warmth
NewsMar.31,2026 -
Discover the Comfort of a USB Heated Blanket for Warmth and Wellness
NewsMar.28,2026














