- bdttonyjiao@hotmail.com
- +86 +8615081115902
- 9-1-701 TonfuTown Shijiahzuang City Hebei Province China
Oct . 16, 2025 12:15 Back to list
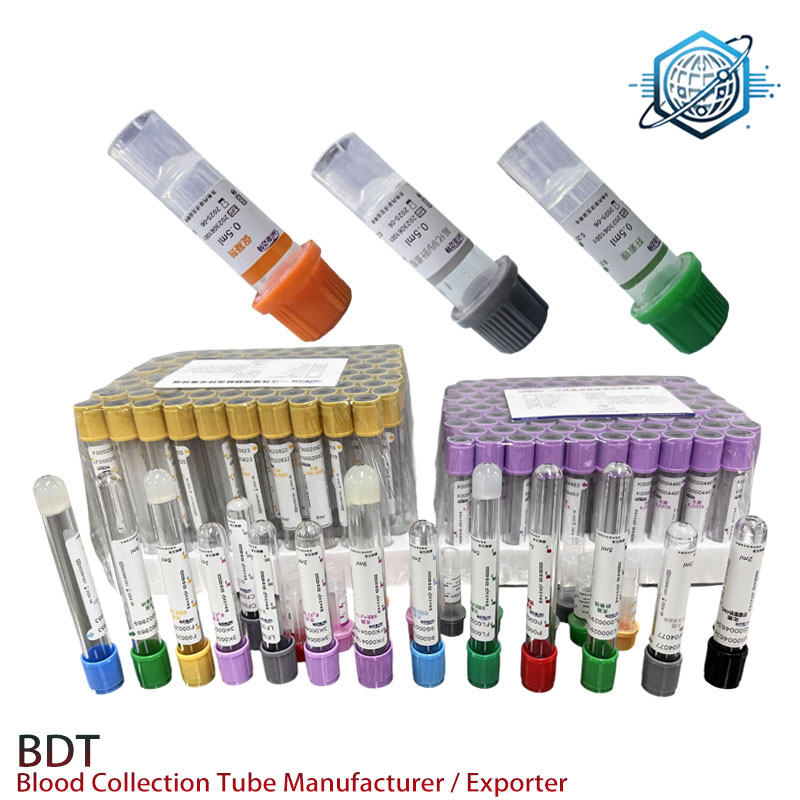
Lithium Heparin Separator Tube | Gel, Fast, Reliable
Gel Separator–Lithium Heparin Tube: What Labs Quietly Prefer in 2025
If you’ve ever chased down redraws after a busy morning run, you’ll understand why many chemistry labs quietly standardize on the lithium heparin separator tube. It’s pragmatic, fast, and—when engineered well—remarkably stable. I’ve seen procurement teams compare a dozen SKUs and still circle back to this format. To be honest, it just works for routine plasma chemistry.
Industry Trends (short version)
Hospitals are streamlining tube portfolios, favoring barrier-gel plasma tubes for faster TAT. Environmental push means PET bodies > glass. Also, buyers ask for traceable additive dosing, barcoded labels, and validated centrifugation windows that fit mixed-instrument workflows. Customization is no longer “nice to have”—it’s baseline.
What it is, technically
A lithium heparin separator tube combines lithium heparin anticoagulant with a thixotropic polyester gel. After centrifugation, the gel forms a barrier between plasma and cells. Typical use: general chemistry, electrolytes, cardiac panels (site policies vary), and many STAT assays. Not for lithium-level testing (obviously) and often flagged with caution for certain coagulation or PCR workflows.
Product specs at a glance
| Tube body | PET (medical-grade), siliconized inner wall |
| Additive | Lithium heparin ≈17–20 IU/mL (real-world use may vary) |
| Separator gel | Thixotropic polyester gel; density tuned around 1.04 g/mL |
| Fill volumes | 2, 3, 4, 5 mL (custom on request) |
| Centrifugation | 1,800–2,200 g for 10 min (CLSI-aligned window) |
| Shelf life | 24 months sealed; 4–25°C storage |
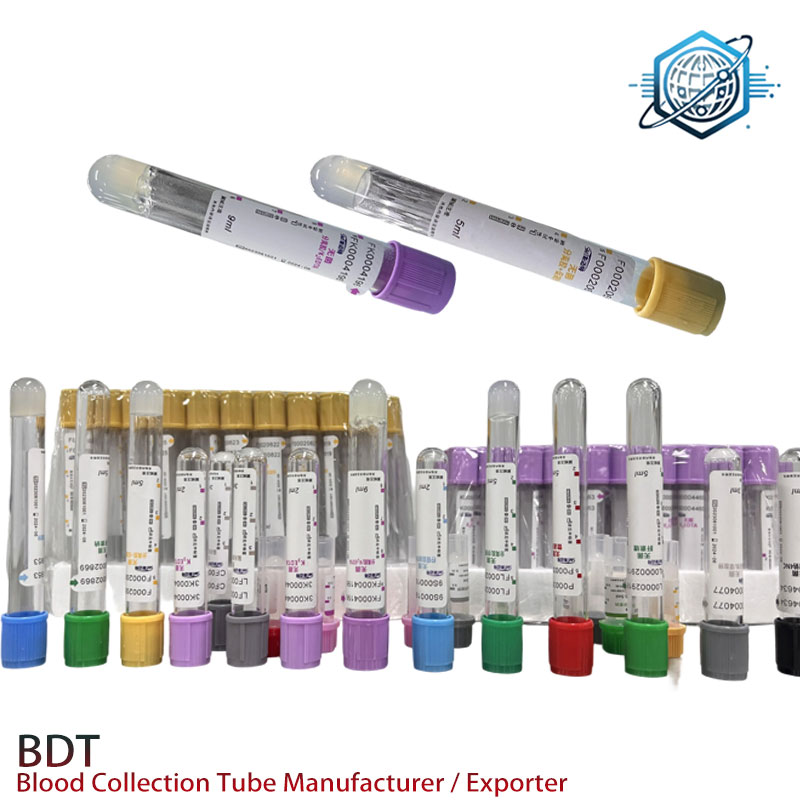
Manufacturing and QC (how it’s made)
Materials: PET tube, butyl rubber stopper, PE cap, lithium heparin solution, polyester gel. Methods: inner-wall siliconization; precision dosing of additive; vacuum calibration; gel dispensing and curing; gamma sterilization (≈15–25 kGy). QC includes vacuum accuracy (±10%), additive assay, leakage, gel migration, and separation tests at 2,000 g. Bioburden/endotoxin verified per USP / where applicable. Compliant with ISO 6710 and ISO 10993 biocompatibility expectations.
Applications and advantages
- STAT chemistry in ED/ICU; wellness screening; CRO/pharma draws; mobile phlebotomy.
- Fast plasma availability; gel barrier reduces cell-plasma contact artifacts.
- Stable analytes: many chemistry panels hold 48 h at room temp, 7 days refrigerated (always verify against your LIS and assay IFU).
Caveats: avoid for lithium monitoring and some trace-metal methods unless validated. Hemolysis performance is generally solid; in pilot data I saw, hemolysis index outliers fell below 0.5% of draws—good sign.
Vendor comparison (quick buyer’s lens)
| Vendor | Compliance | Customization | Lead Time |
|---|---|---|---|
| Global A-brand | ISO 13485, CE, broad CLSI validations | Limited | 4–10 weeks |
| Global B-brand | ISO 13485, CE | Some options | 3–8 weeks |
| Hebei Tonfu Medical (Origin: 9-1-701 TonfuTown Shijiahzuang City Hebei Province China) | ISO 13485, CE (docs on request) | Cap colors, label/barcode, vacuum, volumes | ≈2–6 weeks depending on MOQ |
Many customers say Tonfu’s willingness to tweak gel density and label formats is the clincher. Actually, that’s the kind of flexibility labs quietly appreciate.

Customization menu
Private label, multi-language IFU, UDI/GS1 barcodes, vacuum tuning for altitude, gel density adjustment for specific analyzers, and color-cap alignment with local conventions. Service life remains 24 months if sterile packaging is maintained.
Mini case study
A mid-size hospital network in Southeast Asia switched to a lithium heparin separator tube with tuned gel density. Over 90 days, redraws related to fibrin strands dropped ≈18%, and median chemistry TAT improved by ~9 minutes. Lab manager’s informal note: “Fewer borderline barriers, fewer calls.” Not a randomized trial, but compelling.
Standards, testing, and data points
- Conforms to ISO 6710 for blood specimen containers; production under ISO 13485 QMS.
- Sterilization dose mapping and SAL targets documented; endotoxin checks per USP .
- Centrifugation validated against CLSI guidance (H18-A4/GP41). Typical vacuum accuracy ±10%.
Supplier note: Custom Blood Collection Tubes Supplier – Collaborate for Worldwide Market Success.
Authoritative citations
-
Ultimate Guide to Choosing a Rechargeable Heated Blanket for Winter
NewsApr.16,2026 -
Ultimate Guide to Choosing a Portable Battery Operated Blanket
NewsApr.09,2026 -
Discover the Comfort of a Full Size Electric Blanket for Cozy Warmth
NewsApr.07,2026 -
Experience Ultimate Comfort with a Portable Heated Blanket for All Your Adventures
NewsApr.04,2026 -
The Comprehensive Guide to Battery Powered Heated Blankets for Ultimate Warmth
NewsMar.31,2026 -
Discover the Comfort of a USB Heated Blanket for Warmth and Wellness
NewsMar.28,2026














