- bdttonyjiao@hotmail.com
- +86 +8615081115902
- 9-1-701 TonfuTown Shijiahzuang City Hebei Province China
Oct . 05, 2025 23:30 Back to list
Plain Tube Manufacturer: Premium Materials, Fast Worldwide?
Plain Tube Manufacturer Premium Materials Fast Shipping Worldwide
If you work in diagnostics or phlebotomy, you already know: not all “plain” blood collection tubes are created equal. Our factory in 9-1-701 TonfuTown, Shijiahzuang City, Hebei Province, China has been shipping PET and glass tubes to hospitals and labs on five continents, and—to be honest—the reason people reorder is simple: consistent draw volume, clean serum, and no drama in the centrifuge.
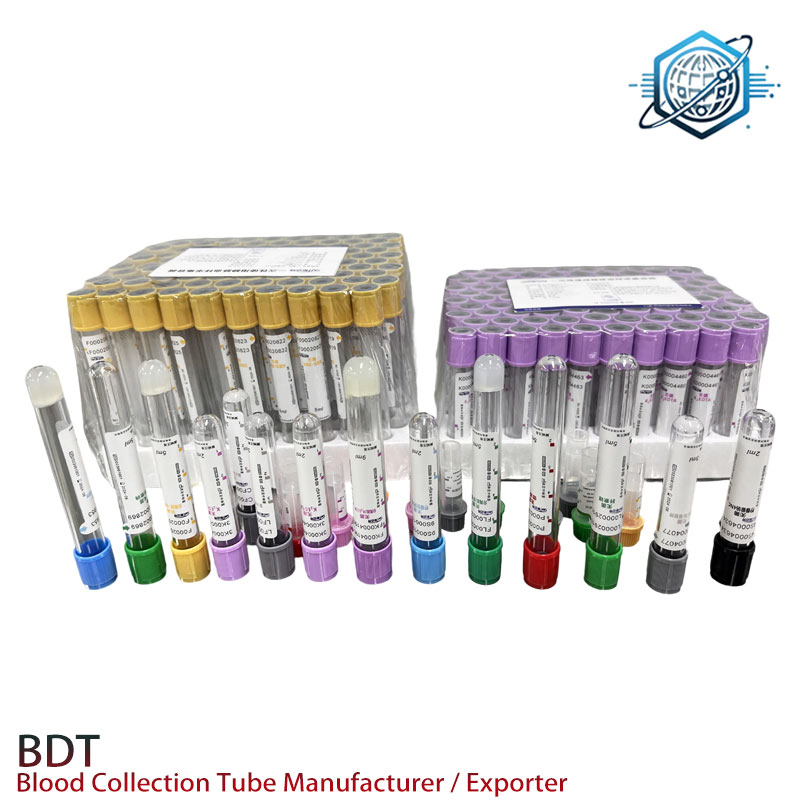
What’s happening in the market
Two trends stand out. First, PET has largely displaced glass in routine venous sampling because it’s lighter and safer; second, labs are asking for faster global replenishment with lower MOQs. We keep venous and capillary formats in stock—yes, really—and ship quickly, which, surprisingly, is still rare.
Product specs at a glance
| Parameter | Specification | Notes |
|---|---|---|
| Materials | PET or Borosilicate Glass; PE/Butyl closure | Medical-grade; latex-free |
| Volume options | 2, 3, 4, 5, 6, 7, 8, 9, 10 mL | Custom fill on request |
| Additives | Plain (no additive) or Clot Activator (silica) | Clot time ≈ 20–60 min (real-world may vary) |
| Vacuum accuracy | ±5% at 23°C | Retention ≥95% at shelf-life end |
| Sterilization | Gamma or EO (SAL ≈ 10⁻⁶) | Inside sterile; exterior clean |
| Centrifuge resistance | Up to 5,000 × g, 10 min | No cap pop, no leakage in QC |
| Shelf life | 24–30 months | Store 4–25°C, dry |

Where these tubes fit
- Clinical chemistry (serum analytes), serology, immunology
- Pharma stability panels and biobanking (plain PET preferred)
- Veterinary labs and field kits—lightweight, shatter-safe
Many customers say they noticed fewer redraws once switching over—seems that consistent vacuum and clean interior finish matter more than people think.
Process and quality flow (short version)
Materials in, components out: PET tube forming → closure molding → surface treatment (optional silicone rinse) → vacuum dosing → assembly in ISO Class 8 cleanroom → 100% visual inspection → lot testing (vacuum, leakage, cap torque, drop/transport, centrifuge) → sterilization → labeling/barcoding → final release under ISO 13485 QMS.
Standards we build against: ISO 6710 (single-use blood specimen container111s), CLSI GP39 for tube performance, IVDR/CE where applicable. Test snapshots: leakage 0/1000 tubes; vacuum retention pass at 24 months; label adhesion pass at 4°C and 40°C stress. That’s the stuff we check, not just talk about.

Vendor comparison (realistic, not marketing)
| Feature | Our Factory | Trader A | Factory B |
|---|---|---|---|
| MOQ | Low (carton-level) | Higher | Medium |
| Lead time | In-stock, ship fast worldwide | Unclear (outsourced) | 2–4 weeks |
| Certs/QMS | ISO 13485, CE/IVD-ready docs | Varies | ISO 9001 |
| Custom barcodes/logo | Yes | Maybe | Limited |
Customization
Cap colors (red, yellow, custom), logo print, pre-barcoding (Code 128/QR), vacuum levels for high altitude sites, and special packouts (sterile inner packs). It’s practical stuff that saves time on your bench.
Mini case study
A biotech in Frankfurt needed 50,000 plain PET tubes, barcoded, in under a week. We shipped in 5 days via air; breakage during transit came in at 0.3% (vs. their previous 1%+). Their lab manager said, “Actually, the steady vacuum saved us more than the freight did.” That’s typical feedback, and I guess that’s why they extended the contract.
Bottom line: if you want Plain Tube Manufacturer Premium Materials Fast Shipping Worldwide performance without a long wait or scary MOQs, you’re in the right place. Venous and capillary formats are in stock; talk to us for samples.
Authoritative references
- ISO 6710:2017 Single-use container111s for venous whole blood specimen collection.
- CLSI GP39 (latest): Tubes and Additives for Venous and Capillary Blood Specimen Collection.
- ISO 13485:2016 Medical devices—Quality management systems—Requirements for regulatory purposes.
- Regulation (EU) 2017/746 (IVDR) on in vitro diagnostic medical devices.
- ISO 11137-1:2006 Sterilization of health care products—Radiation—Requirements.
-
Ultimate Guide to Choosing a Rechargeable Heated Blanket for Winter
NewsApr.16,2026 -
Ultimate Guide to Choosing a Portable Battery Operated Blanket
NewsApr.09,2026 -
Discover the Comfort of a Full Size Electric Blanket for Cozy Warmth
NewsApr.07,2026 -
Experience Ultimate Comfort with a Portable Heated Blanket for All Your Adventures
NewsApr.04,2026 -
The Comprehensive Guide to Battery Powered Heated Blankets for Ultimate Warmth
NewsMar.31,2026 -
Discover the Comfort of a USB Heated Blanket for Warmth and Wellness
NewsMar.28,2026














