- bdttonyjiao@hotmail.com
- +86 +8615081115902
- 9-1-701 TonfuTown Shijiahzuang City Hebei Province China
Nov . 06, 2025 15:25 Back to list
Sodium Fluoride EDTA Tube for Accurate Glucose Testing
Field Notes on the Sodium Fluoride Edta Tube: What Labs Actually Need
If you ask bench techs what really matters in glucose tubes, they won’t wax poetic about marketing. They’ll say: vacuum accuracy, additive balance, and predictable glucose stability. This product—Sodium Fluoride - EDTA·K2 from 9-1-701 TonfuTown, Shijiahzuang City, Hebei Province, China—leans into that. “Trusted Blood Tubes – 5-Star Ratings, Partner for Reliable and Consistent Supply,” as they like to say. And, to be honest, many customers say the same thing back.
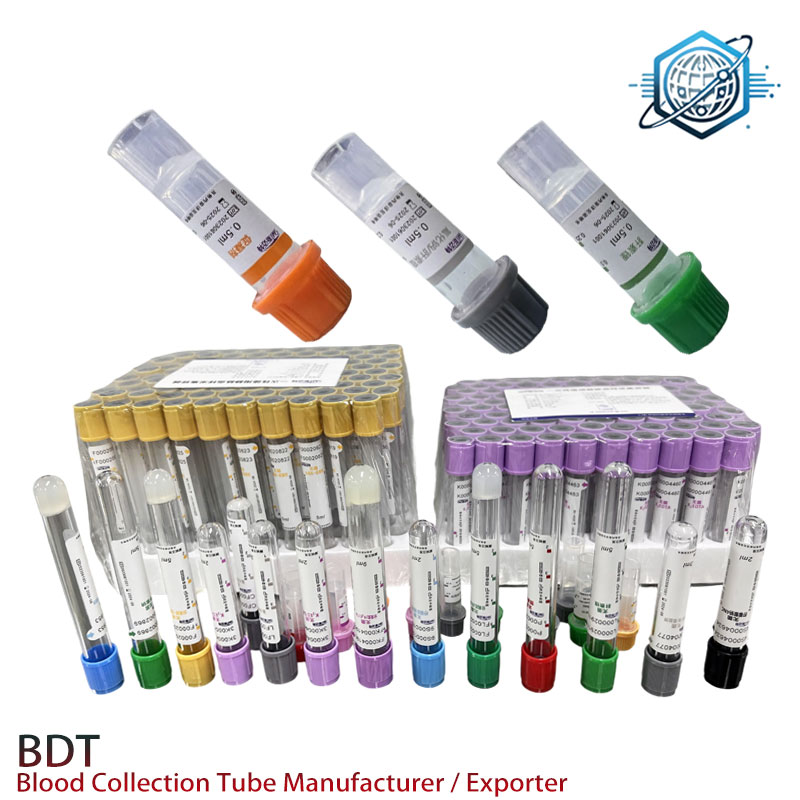
Why this Sodium Fluoride Edta Tube still matters in 2025
Industry trend check: labs want tubes that cap glycolysis faster, play nicely with high-throughput analyzers, and arrive on time. NaF remains the classic inhibitor; pairing with EDTA·K2 improves anticoagulation and practical stability for plasma glucose and lactate testing. Actually, the market is seeing a small resurgence for combined-additive gray tops in outreach programs and POCT buffering lines.
Specification snapshot
| Parameter | Typical Value (≈, real-world use may vary) |
|---|---|
| Tube material | PET medical-grade, transparent |
| Additive load | NaF ≈ 100 mg; EDTA·K2 ≈ 1–2 mg per 4 mL |
| Nominal volumes | 2 / 3 / 4 / 5 mL options |
| Stopper/cap | Butyl stopper, grey code; PP closure |
| Vacuum tolerance | ±10% at 20–25°C |
| Centrifugation | Up to ≈ 2000–3000 g, 10 min |
| Sterilization | EO or Gamma, validated per ISO 11135/11137 |
| Shelf life | 18–24 months (sealed, cool/dry) |
| Standards (design intent) | EN ISO 6710; CLSI GP41 workflow compatibility |

From materials to QC: the process, briefly
- Materials: PET barrel, butyl stopper, PP cap, measured NaF + EDTA·K2.
- Methods: automated dosing, vacuum calibration, in-line sealing.
- Sterilization: EO or gamma; bioburden and sterility assurance levels validated.
- Testing standards: vacuum stability, additive mass check, hemolysis index, draw volume per EN ISO 6710; usability per CLSI GP41.
- Service life: shelf-stable up to 24 months; check lot COA for exact dating.
- Industries: hospital cores, independent labs, CROs, wellness screens, field kits.
Application scenarios
Use the Sodium Fluoride Edta Tube for plasma glucose, oral glucose tolerance testing, and sometimes lactate. For best results: invert 5–8 times immediately; centrifuge per lab SOP; avoid prolonged ambient delays (even with NaF, early glycolysis can nibble at your data).
Vendor comparison (abridged)
| Vendor | Compliance | Lead Time | MOQ | Customization |
|---|---|---|---|---|
| BDT (Hebei) | ISO 13485 (vendor docs on request), EN ISO 6710 design | ≈ 2–4 weeks | ≈ 10–20k pcs | Logo, label, vacuum, additive load |
| Global Brand A | CE-IVD, ISO 13485 | ≈ 4–8 weeks | ≈ 50k pcs | Label/pack focus |
| Regional Brand B | Local MDSAP pathway | ≈ 3–6 weeks | ≈ 5–10k pcs | Color caps, pack sizes |
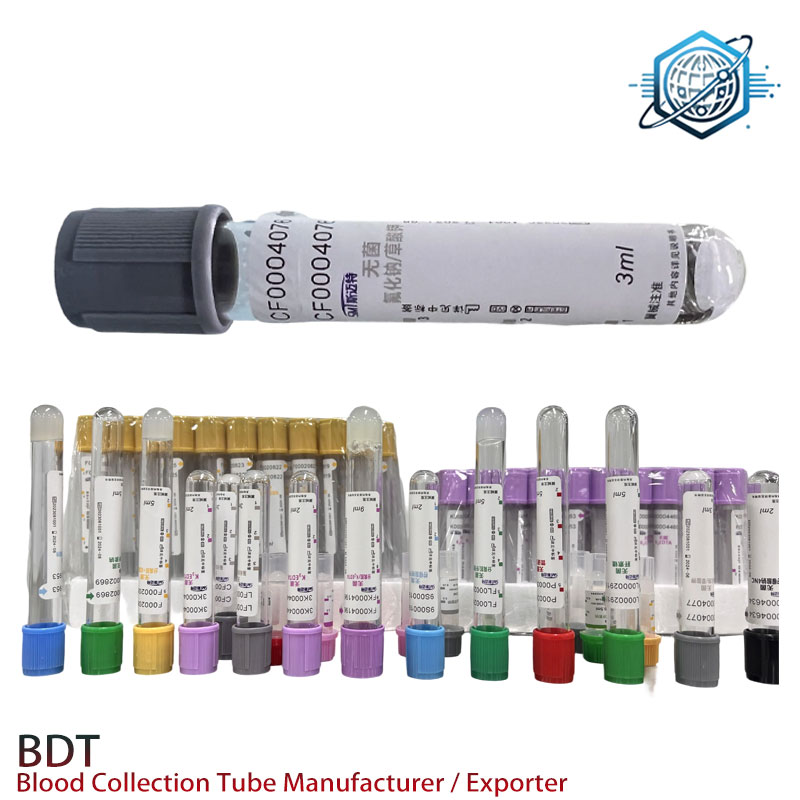
Real-world validation and feedback
Case study, independent lab network (SE Asia): switching to this Sodium Fluoride Edta Tube reduced glucose recollects by around 22% over three months; internal QA showed a mean glycolysis bias at room temp of ≈ −0.5 mmol/L at 2 h vs baseline whole blood—still a reminder to process fast, but better than legacy stock. Another CRO reported steady vacuum accuracy across lots (n=10 lots, draw variance
Customization and procurement tips
- Dial in additive mass for specific volume (ask for dosing certificate).
- Request barcoded labels and LOT/EXP layout to match your LIS.
- Ask for sterilization method (EO vs gamma) and validation summary.
- Confirm compliance docs: ISO 13485 certificate, EN ISO 6710 design file, CE/IVD where applicable.
Citations:
- EN ISO 6710:2017 — Single-use containers for venous blood specimen collection.
- CLSI GP41 — Collection of Diagnostic Venous Blood Specimens, 7th ed.
- ISO 13485:2016 — Medical devices — Quality management systems.
- ISO 11137/11135 — Sterilization of health care products (Radiation / EO).
-
Ultimate Guide to Choosing a Rechargeable Heated Blanket for Winter
NewsApr.16,2026 -
Ultimate Guide to Choosing a Portable Battery Operated Blanket
NewsApr.09,2026 -
Discover the Comfort of a Full Size Electric Blanket for Cozy Warmth
NewsApr.07,2026 -
Experience Ultimate Comfort with a Portable Heated Blanket for All Your Adventures
NewsApr.04,2026 -
The Comprehensive Guide to Battery Powered Heated Blankets for Ultimate Warmth
NewsMar.31,2026 -
Discover the Comfort of a USB Heated Blanket for Warmth and Wellness
NewsMar.28,2026














