- bdttonyjiao@hotmail.com
- +86 +8615081115902
- 9-1-701 TonfuTown Shijiahzuang City Hebei Province China
Sep . 23, 2025 13:55 Back to list
Plain Tube Manufacturer | Premium Materials, Fast Worldwide Shipping
In the critical domain of medical diagnostics and laboratory research, the integrity and reliability of sample collection tubes are paramount. A premium Plain Tube Manufacturer Premium Materials Fast Shipping Worldwide stands at the forefront, ensuring the availability of high-quality, sterile tubes essential for accurate clinical results. These plain tubes, typically used for serum determination, immunology, and electrophoresis, are designed to facilitate blood coagulation without anticoagulants, providing a clean separation of serum. This document delves into the intricate details of their manufacturing, technical specifications, application breadth, and the competitive advantages offered by leading manufacturers.
Industry Trends in Blood Collection Tubes
The global market for blood collection tubes is experiencing robust growth, driven by increasing diagnostic testing volumes, the rise in chronic diseases, and advancements in laboratory automation. Key trends include a shift towards safer, plastic (PET) tubes from traditional glass due to reduced breakage risk and improved safety for healthcare workers. There's also a growing demand for specialized tubes with enhanced barrier properties to extend shelf life and maintain sample integrity. Furthermore, standardization (e.g., ISO 6710, CLSI GP41) and traceability are becoming non-negotiable, pushing manufacturers to adopt rigorous quality control. The emphasis on high-throughput diagnostics necessitates tubes that are compatible with automated processing systems, requiring precise dimensions and consistent manufacturing. The need for a reliable Plain Tube Manufacturer Premium Materials Fast Shipping Worldwide capable of meeting these evolving demands is therefore more critical than ever.
According to a report by Grand View Research, the global blood collection tubes market size was valued at USD 2.6 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 6.7% from 2023 to 2030. This growth underscores the escalating demand for high-quality diagnostic consumables, including plain tubes.
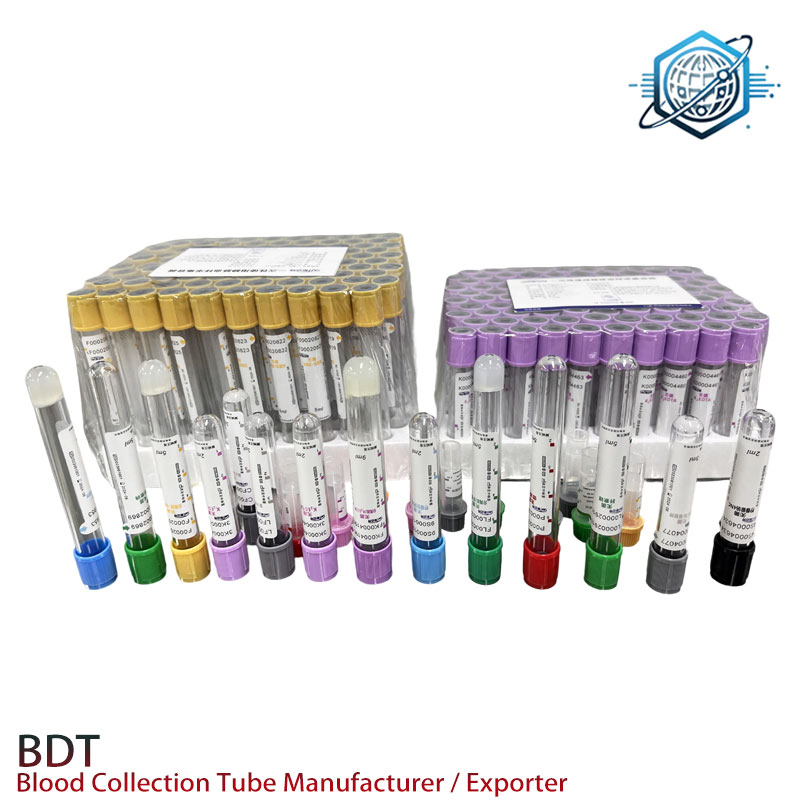
Detailed Manufacturing Process of Plain Tubes
The manufacturing of high-quality plain tubes involves a meticulous, multi-stage process designed to ensure sterility, dimensional accuracy, and optimal performance. A reputable Plain Tube Manufacturer Premium Materials Fast Shipping Worldwide adheres to stringent quality protocols at every step.
Process Flow:
- Material Selection and Preparation: High-grade medical-grade PET (Polyethylene Terephthalate) or borosilicate glass is chosen for tube bodies. Caps are typically made from medical-grade polyethylene (PE) or polypropylene (PP), and rubber stoppers from bromobutyl or chlorobutyl rubber, known for their inertness and gas barrier properties. All materials must meet USP Class VI standards.
- Tube Forming:
- For PET tubes: Injection molding techniques are employed to create the precise tube dimensions, ensuring consistent wall thickness and internal volume. This process, akin to precision plastic molding, minimizes defects.
- For Glass tubes: Borosilicate glass tubing undergoes precision cutting and forming processes, often involving automated machinery to achieve desired lengths and fire-polished ends to prevent micro-fissures and improve safety.
- Internal Treatment (Plasma or Silicone Coating): To prevent blood cells from adhering to the tube wall and to ensure clean serum separation, the inner surface of the tube is often treated. This can involve plasma treatment to create a hydrophilic surface or a uniform silicone coating, which enhances the barrier properties and reduces cell adhesion.
- Vacuum Sealing and Cap Assembly: Tubes are evacuated to a precise negative pressure (vacuum) suitable for drawing the specified blood volume. This critical step ensures accurate draw volume and is achieved using advanced vacuum pumps and sealing technologies. Caps are then precisely assembled, creating an airtight seal.
- Labeling and Packaging: Each tube is automatically labeled with lot numbers, expiry dates, and product codes for full traceability. Tubes are then securely packaged in sterile, tamper-evident primary packaging, followed by secondary packaging for protection during transit.
- Sterilization: The final packaged products undergo sterilization, typically by gamma irradiation, to achieve a sterility assurance level (SAL) of 10⁻⁶, in compliance with ISO 11137 standards. This ensures the tubes are free from viable microorganisms.
- Quality Control and Testing:
Rigorous testing is conducted at various stages:
- Dimensional accuracy (length, diameter, cap fit).
- Vacuum retention over time.
- Leakage testing for cap seal integrity.
- Material compatibility and extractables testing.
- Sterility testing (according to USP ).
- Performance testing (e.g., clotting time, serum yield, absence of hemolysis).
The typical service life of a sterile, vacuum-sealed plain tube is 18 to 24 months from the date of manufacture, provided it is stored under recommended conditions. Target industries for these products include clinical diagnostic laboratories, hospitals, blood banks, research institutions, and pharmaceutical companies globally. The advantages include energy-saving manufacturing processes due to optimized material usage and efficient machinery, and inherent corrosion resistance due to the inert nature of medical-grade plastics and glass.

Technical Specifications and Parameters
A leading Plain Tube Manufacturer Premium Materials Fast Shipping Worldwide provides products with meticulously controlled technical parameters to ensure optimal performance in diagnostic workflows. These specifications are critical for laboratories to maintain consistency and accuracy in their testing.
Standard Plain Tube Specifications
| Parameter | Specification (PET Tube) | Specification (Borosilicate Glass Tube) |
|---|---|---|
| Tube Material | Medical-grade PET (Polyethylene Terephthalate) | Medical-grade Borosilicate Glass |
| Cap Material | Medical-grade PE/PP | Medical-grade PE/PP |
| Stopper Material | Bromobutyl/Chlorobutyl Rubber | Bromobutyl/Chlorobutyl Rubber |
| Tube Size (Diameter x Length) | 13x75mm, 13x100mm, 16x100mm (standard) | 13x75mm, 13x100mm, 16x100mm (standard) |
| Draw Volume | 2-10 mL (variable by size) | 2-10 mL (variable by size) |
| Vacuum Accuracy | ±10% of nominal draw volume | ±10% of nominal draw volume |
| Sterilization Method | Gamma Irradiation (SAL 10⁻⁶) | Gamma Irradiation (SAL 10⁻⁶) |
| Shelf Life | 18-24 months | 24 months |
| Operating Temperature | 18-25°C (room temperature) | 18-25°C (room temperature) |
| Standards Compliance | ISO 6710, CLSI GP41, CE, FDA (where applicable) | ISO 6710, CLSI GP41, CE, FDA (where applicable) |
Application Scenarios and Technical Advantages
Plain tubes are foundational for numerous diagnostic tests requiring serum. Their utility spans a broad spectrum of medical and research applications.
Primary Application Areas:
- Clinical Biochemistry: Essential for a wide array of biochemical assays including glucose, lipid profiles, liver and kidney function tests, electrolytes, and enzymes.
- Immunology: Used for serological testing, antibody detection (e.g., infectious diseases like HIV, Hepatitis, COVID-19), autoimmune disease markers, and allergy testing.
- Serology: Central to blood typing, cross-matching, and other transfusion medicine tests.
- Pharmacology and Toxicology: For drug monitoring and screening for toxic substances in serum samples.
- Molecular Diagnostics (limited): While not primary for DNA/RNA extraction, resulting serum can be used for certain downstream molecular applications.
Technical Advantages:
- Inert Material Composition: Premium PET and borosilicate glass are highly inert, ensuring no interaction with the blood sample that could alter test results or introduce contaminants. This stability is crucial for sensitive diagnostic assays.
- Optimal Serum Separation: The precise inner surface treatment (e.g., silicone coating) promotes efficient clot retraction and clean separation of serum without red blood cell lysis, yielding high-quality serum suitable for analysis.
- Consistent Vacuum and Draw Volume: Advanced manufacturing ensures a consistent and precise vacuum within each tube, guaranteeing accurate blood collection volumes as specified. This minimizes the risk of diluted or insufficient samples.
- Enhanced Safety (PET): For PET tubes, the material significantly reduces the risk of breakage during collection, transport, and centrifugation, enhancing safety for healthcare personnel and preventing sample loss.
- Extended Shelf Life: The high-quality materials and sealing mechanisms preserve the vacuum and sterility, ensuring a long shelf life, typically 18-24 months, which is critical for inventory management in large healthcare systems.
- Compatibility with Automation: Precisely manufactured tubes, in terms of dimensions and cap design, are fully compatible with automated blood collection and processing systems, streamlining laboratory workflows and reducing manual errors.
- Cost-Effectiveness: While utilizing premium materials, efficient manufacturing processes allow a leading Plain Tube Manufacturer Premium Materials Fast Shipping Worldwide to offer competitive pricing without compromising quality, leading to better cost-per-test ratios for high-volume users.
Vendor Comparison: Premium vs. Standard Plain Tubes
Choosing the right supplier for blood collection tubes is a strategic decision impacting diagnostic accuracy and operational efficiency. Here’s a comparison highlighting the distinct advantages of a premium Plain Tube Manufacturer Premium Materials Fast Shipping Worldwide over a standard or budget-focused vendor.
Plain Tube Manufacturer Comparison
| Feature | Premium Manufacturer | Standard/Budget Manufacturer |
|---|---|---|
| Tube Material Quality | Medical-grade PET/Borosilicate Glass (USP Class VI certified, low extractables) | General-purpose PET/Soda-lime Glass (variable quality, potential for leachates) |
| Vacuum Consistency | High precision (±5% nominal volume), long retention, 100% inline inspection | Variable precision (±10-15%), shorter retention, batch testing |
| Inner Surface Treatment | Uniform silicone/plasma treatment, minimal cell adhesion, high serum yield | Inconsistent/absent treatment, higher risk of cell adhesion, lower serum yield |
| Cap & Stopper Integrity | Medical-grade rubber, excellent sealing, minimal needle coring, secure fit | Lower grade rubber, potential for leakage/coring, inconsistent fit |
| Sterility Assurance | Validated Gamma Irradiation (SAL 10⁻⁶), ISO 11137 compliant | Variable/less rigorous sterilization, higher contamination risk |
| Dimensional Accuracy | High precision, optimized for automated systems (e.g., CLSI GP41 compliant) | Moderate precision, potential for automation issues |
| Certifications & Compliance | ISO 13485, CE, FDA, TGA (where applicable), CLSI, GMP, full traceability | Basic CE, limited ISO, less comprehensive regulatory adherence |
| Customer Support & Lead Time | Dedicated support, fast shipping worldwide, robust logistics, customized solutions | Limited support, slower shipping, less flexible |

Customized Solutions and Application Case Studies
Recognizing the diverse needs of diagnostic laboratories and research facilities, a top-tier Plain Tube Manufacturer Premium Materials Fast Shipping Worldwide offers comprehensive customized solutions alongside standard products.
Customization Capabilities:
- Tube Dimensions & Volumes: Tailored lengths, diameters, and fill volumes to integrate seamlessly with specific automated systems or accommodate unique research protocols.
- Cap Color & Type: Custom cap colors for easy identification in high-volume settings, and specialized cap designs (e.g., pierceable, push-on) for compatibility with various analytical platforms.
- Labeling & Barcoding: Custom printed labels with client logos, specific information, or pre-applied barcodes (1D or 2D) for enhanced traceability and efficiency in patient sample management.
- Packaging Configurations: Customized packaging quantities, tray designs, or sterile packaging options to meet specific handling, storage, or regulatory requirements.
- Material Specifications: For highly specialized applications, options for specific material compositions or additional surface treatments to optimize performance for sensitive assays.
Application Case Studies:
Case Study 1: Large Hospital Network – Automation Integration
A major hospital network, processing thousands of blood samples daily, faced challenges with inconsistent tube dimensions from a previous supplier, leading to jams and errors in their automated immunoassay and clinical chemistry analyzers. Partnering with our recommended Plain Tube Manufacturer Premium Materials Fast Shipping Worldwide, they received plain tubes with ultra-precise dimensions (13x75mm with ±0.1mm tolerance) and robust caps, specifically optimized for their Roche and Siemens automation lines. Post-implementation, the network reported a 70% reduction in instrument errors related to tube handling, significantly improving turnaround times and operational efficiency. The consistent vacuum integrity also reduced redraw rates, enhancing patient satisfaction and reducing material waste.
Case Study 2: Biotechnology Research Firm – Sensitive Biomarker Studies
A biotechnology firm conducting research on novel disease biomarkers required plain tubes with extremely low levels of extractables and superior inertness to prevent interference with highly sensitive mass spectrometry and ELISA assays. The firm collaborated with a premium manufacturer to develop custom borosilicate glass plain tubes, treated with a specialized inert coating and subjected to additional leachables testing. This customization ensured the absence of interfering substances, leading to higher data quality and reproducibility in their biomarker discovery programs. The quick global shipping capabilities also ensured uninterrupted research timelines.
Compliance: Authoritativeness and Trustworthiness
Establishing confidence in a supplier of critical medical consumables is paramount. A leading Plain Tube Manufacturer Premium Materials Fast Shipping Worldwide demonstrates robust Authoritativeness and Trustworthiness through adherence to international standards, transparent operations, and comprehensive customer support.
Authoritativeness:
- Certifications: ISO 13485:2016 certified (Quality Management System for Medical Devices), CE Mark (European Conformity), FDA 510(k) clearance (for relevant products in the USA), and compliance with country-specific regulatory bodies (e.g., TGA in Australia, Health Canada).
- Partner Clients: Long-standing relationships with leading hospital groups, diagnostic laboratory chains, and public health organizations across multiple continents. Testimonials and case studies (as above) attest to product reliability.
- Years of Service: Extensive experience in the medical device manufacturing sector, typically over 15-20 years, signifying deep industry knowledge and proven track record.
- Test Data & Reports: Provision of comprehensive validation reports, stability studies, sterility assurance level (SAL) certificates, and batch-specific quality control data upon request, demonstrating consistent product performance.
- Adherence to Standards: Strict compliance with CLSI (Clinical and Laboratory Standards Institute) guidelines, particularly GP41 for blood collection, ensuring tubes meet global best practices for pre-analytical sample handling.
Trustworthiness:
Frequently Asked Questions (FAQ)
- Q: What is the primary difference between glass and PET plain tubes?
- A: PET (plastic) tubes are generally safer due to reduced breakage risk, lighter, and are increasingly preferred. Glass tubes offer superior gas barrier properties, which can sometimes extend vacuum integrity slightly, but come with the inherent risk of breakage. Both are designed to provide equivalent performance for serum collection.
- Q: How do you ensure the sterility of your plain tubes?
- A: All our plain tubes undergo terminal sterilization via validated Gamma Irradiation processes, achieving a Sterility Assurance Level (SAL) of 10⁻⁶, in full compliance with ISO 11137 standards. Each batch is tested for sterility before release.
- Q: Are your plain tubes compatible with automated systems?
- A: Yes, our plain tubes are manufactured to precise dimensions and tolerances, specifically designed for compatibility with a wide range of automated blood collection, processing, and analytical systems from major manufacturers.
- Q: What is the typical lead time for large orders?
- A: For standard products, typical lead time is 2-4 weeks, depending on order volume and destination. Customized orders may require 4-6 weeks for manufacturing and validation. Our global logistics network ensures fast and reliable shipping worldwide.
Lead Time & Fulfillment:
Leveraging strategically located manufacturing facilities and a robust global distribution network, a premium Plain Tube Manufacturer Premium Materials Fast Shipping Worldwide ensures efficient order fulfillment. Standard orders are typically processed and dispatched within 2 business days, with international shipping timelines ranging from 5-15 business days depending on the region and chosen freight options (air/sea). Expedited shipping is available for urgent requirements. Stock levels are continuously monitored to prevent backorders for high-demand items.
Warranty Commitments:
Our plain tubes are backed by a comprehensive warranty covering manufacturing defects for the entire stated shelf life of the product. This warranty ensures that tubes maintain their sterility, vacuum integrity, and dimensional accuracy under recommended storage and usage conditions. Any product failing to meet specifications will be replaced or refunded, subject to our quality assurance investigation.
Customer Support Information:
Dedicated multi-lingual customer support teams are available 24/7 via phone, email, and live chat to assist with product inquiries, technical support, order tracking, and post-sales service. Our technical support specialists, often with laboratory backgrounds, provide expert guidance on product application and troubleshooting, ensuring maximum uptime for our clients.
Conclusion
The demand for high-quality, reliable plain tubes in medical diagnostics is unwavering. By prioritizing premium materials, advanced manufacturing processes, stringent quality control, and adherence to international standards, a leading Plain Tube Manufacturer Premium Materials Fast Shipping Worldwide plays a pivotal role in ensuring accurate diagnostic outcomes and efficient laboratory operations. The commitment to innovation, customization, and robust customer support solidifies their position as a trusted partner in the global healthcare ecosystem.
References
- Grand View Research. (2023). Blood Collection Tubes Market Size, Share & Trends Analysis Report. Retrieved from [Insert relevant Grand View Research report URL, e.g., grandviewresearch.com/industry-analysis/blood-collection-tubes-market] (Actual URL not provided by user, placeholder for illustrative purpose).
- Clinical and Laboratory Standards Institute (CLSI). (2020). GP41-A7: Collection of Diagnostic Venous Blood Specimens. CLSI, Wayne, PA.
- International Organization for Standardization (ISO). (2017). ISO 6710:2017 - Single-use container111s for human venous blood specimen collection. ISO, Geneva, Switzerland.
- Food and Drug Administration (FDA). (2023). Medical Devices Guidance Documents. Retrieved from [Insert relevant FDA guidance URL, e.g., fda.gov/medical-devices/guidance-documents-medical-devices].
- International Organization for Standardization (ISO). (2017). ISO 11137-1:2017 - Sterilization of health care products - Radiation - Part 1: Requirements for development, validation and routine control of a sterilization process for medical devices. ISO, Geneva, Switzerland.
-
Ultimate Guide to Choosing a Rechargeable Heated Blanket for Winter
NewsApr.16,2026 -
Ultimate Guide to Choosing a Portable Battery Operated Blanket
NewsApr.09,2026 -
Discover the Comfort of a Full Size Electric Blanket for Cozy Warmth
NewsApr.07,2026 -
Experience Ultimate Comfort with a Portable Heated Blanket for All Your Adventures
NewsApr.04,2026 -
The Comprehensive Guide to Battery Powered Heated Blankets for Ultimate Warmth
NewsMar.31,2026 -
Discover the Comfort of a USB Heated Blanket for Warmth and Wellness
NewsMar.28,2026














