- bdttonyjiao@hotmail.com
- +86 +8615081115902
- 9-1-701 TonfuTown Shijiahzuang City Hebei Province China
Oct . 28, 2025 16:00 Back to list
K3 EDTA Tube Price: Bulk Discounts, CE/ISO, Fast Shipping
What affects K3 Edta Tube Price in 2025? A practical buyer’s guide
If you source lavender-top tubes for hematology, you’ve probably noticed prices wobble more than you’d expect. Resin markets, sterilization capacity, and even shipping lanes nudge costs up or down. I’ve spent time with procurement teams and factory folks in Hebei and beyond; here’s the short version: consistency and compliance often matter more than a few cents. Still, the K3 Edta Tube Price is a fair question—and the answer isn’t just a number; it’s a stack of specs and process controls.
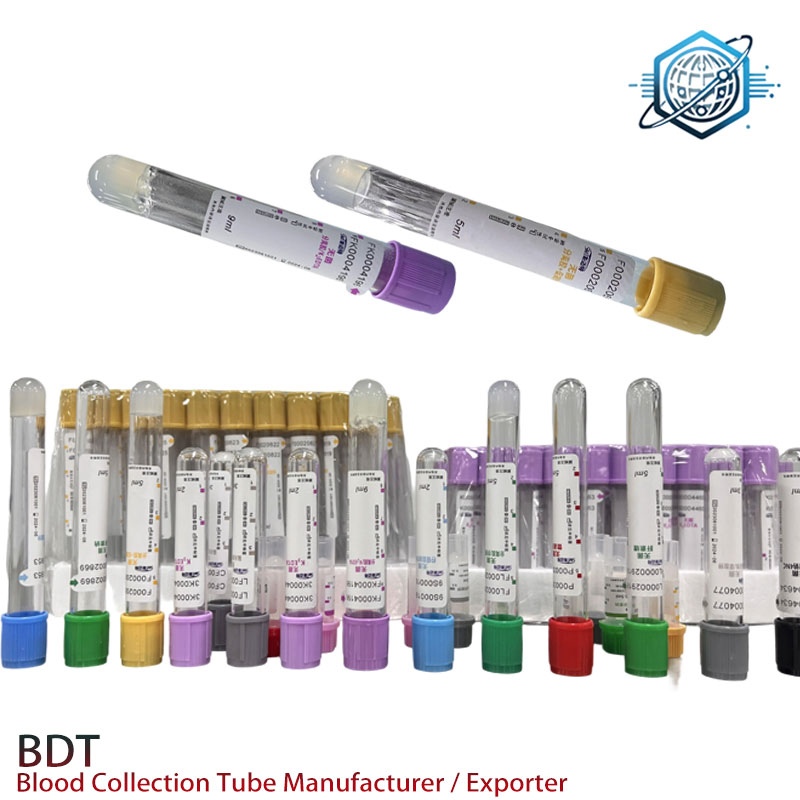
Product snapshot: EDTAK3 tubes from Hebei
Product: EDTAK3 | Origin: 9-1-701 TonfuTown, Shijiahzuang City, Hebei, China. The maker positions itself as a leading venous & capillary blood collection tube manufacturer—global importers wanted, as they say. In practice, that means private-label options, flexible MOQs, and surprisingly quick lead times when the resin supply behaves.
Where these tubes are used
- Routine CBC, WBC differentials, reticulocytes
- HbA1c, malaria smears, many PCR workflows (EDTA chelates Mg²⁺, helpful for DNA integrity)
- Hospital core labs, independent diagnostics, donor screening (non-serology)
Why the K3 Edta Tube Price fluctuates
Key drivers: PET vs glass choice, additive dosing precision, sterilization route (EO vs gamma), vacuum accuracy, quality certifications (ISO 13485, CE-IVD), and packaging/barcoding customization. Freight and carton yield—oddly—matter a lot too.
Key specifications (field-proven)
| Parameter | Spec (≈, real-world use may vary) |
| Tube material | PET or glass |
| Additive | K3EDTA spray-dried, 1.5–2.2 mg/mL blood |
| Volumes | 2, 3, 4, 5, 6 mL options |
| Vacuum accuracy | ±10% draw, retention ≥96 h @ 20–25°C |
| Closure | Butyl rubber, lavender cap, pierceable |
| Sterilization | EO or Gamma (ISO 11135/11137) |
| Shelf life | 18–24 months sealed |
| Compliance | ISO 13485, ISO 6710, CE-IVD; QSR 21 CFR 820 aligned |
| Test data (typ.) | MCV drift ≤1% @24 h (4–8°C); hemolysis index ≤10; endotoxin |
Process flow that impacts price
Materials matter—pharma-grade PET, medical butyl rubber, and consistent EDTA coating. Methods: ultrasonic washing, silicone surface treatment, controlled EDTA spray, vacuum dosing, inline vision checks. Testing: vacuum/volume verification, additive assay, sterility and bioburden, dye leak, cap torque. Service life hinges on seal integrity and storage (4–25°C, dry).
Vendor comparison (indicative)
| Vendor | MOQ | Lead Time | Vacuum stability (30 d) | Certs | USD/1k (ex-works) |
| EDTAK3 (Hebei) | ≈10k | 2–4 wks | ≥95% retained | ISO 13485, CE-IVD | $18–28 (PET) |
| OEM A (SEA) | ≈20k | 3–6 wks | ≈93% | ISO 13485 | $20–32 |
| EU Brand (import) | stock | 1–2 wks | ≥96% | IVDR, ISO 13485 | $35–55 |
Note: ranges are indicative; lane, resin, sterilization, and packaging can swing the K3 Edta Tube Price.
Customization that labs actually use
- Private label, lavender shade matching, multilingual IFUs
- Barcode: Code 128/GS1, serialized lots
- Additive tuning for pediatric micro-volumes

Mini case: fewer recollects, happier night shift
A mid-size hospital lab in the Gulf swapped to EDTAK3 PET tubes with tighter vacuum control. Over 60 days, redraws due to underfill dropped ≈23%, platelet clumping flags fell slightly (techs said “noticeably calmer”). Cost per test nudged down even though unit price was mid-range. That’s the hidden part of K3 Edta Tube Price: total cost of quality.
Standards to check before you buy
- ISO 6710 container compliance and labeling
- CLSI GP41 venipuncture sequence and tube handling
- Manufacturer QMS: ISO 13485; alignment to FDA 21 CFR 820
- Validation: CBC stability up to 24 h (MCV/MPV drift), endotoxin, sterility
References
- ISO 6710:2017 – Single-use containers for venous blood specimen collection.
- CLSI GP41 – Collection of Diagnostic Venous Blood Specimens, current ed.
- ISO 13485:2016 – Medical devices QMS requirements.
- FDA 21 CFR Part 820 – Quality System Regulation.
- WHO – WHO guidelines on drawing blood: best practices in phlebotomy.
-
Ultimate Guide to Choosing a Rechargeable Heated Blanket for Winter
NewsApr.16,2026 -
Ultimate Guide to Choosing a Portable Battery Operated Blanket
NewsApr.09,2026 -
Discover the Comfort of a Full Size Electric Blanket for Cozy Warmth
NewsApr.07,2026 -
Experience Ultimate Comfort with a Portable Heated Blanket for All Your Adventures
NewsApr.04,2026 -
The Comprehensive Guide to Battery Powered Heated Blankets for Ultimate Warmth
NewsMar.31,2026 -
Discover the Comfort of a USB Heated Blanket for Warmth and Wellness
NewsMar.28,2026














