- bdttonyjiao@hotmail.com
- +86 +8615081115902
- 9-1-701 TonfuTown Shijiahzuang City Hebei Province China
Aug . 30, 2025 05:00 Back to list
Premium EDTA Vacuum Blood Collection Tubes for Lab Accuracy
The Critical Role of Advanced Blood Collection Technologies
In the intricate landscape of clinical diagnostics and medical research, the integrity and quality of blood samples are paramount. Precision in sample collection directly influences the accuracy and reliability of diagnostic results, making the choice of collection devices a critical factor. Among these, edta vacuum blood collection tubes stand as an indispensable component for a vast array of hematological and molecular tests. Their design and formulation are specifically engineered to preserve cellular components and nucleic acids, preventing coagulation while maintaining cellular morphology, which is crucial for accurate analysis.
Industry Trends and Market Dynamics
The market for blood collection tubes is experiencing continuous evolution, driven by advancements in diagnostic methodologies, increasing demand for point-of-care testing, and a heightened focus on patient and healthcare worker safety. Key trends include the transition towards plastic tubes for enhanced safety, miniaturization for pediatric and specialized applications (leading to increased demand for small edta tubes), and the integration of smart technologies for sample tracking and data management. Moreover, the emphasis on pre-analytical phase error reduction is pushing manufacturers to develop more standardized and robust products. Innovations in additive formulations, such as optimized potassium edta tube concentrations and spray-drying techniques, further enhance sample stability and extend viable testing windows. The global market is projected to grow significantly, reflecting the escalating need for accurate and efficient diagnostic solutions across various healthcare settings, from large reference laboratories to local clinics.
Demand for high-quality vacutainer edta tubes (a widely recognized brand example) and generic equivalents continues to rise, especially with the expansion of genetic testing, personalized medicine, and routine health screenings. Ensuring a consistent supply of these essential laboratory consumables, which meet stringent international standards, is critical for maintaining diagnostic efficiency worldwide.
Manufacturing Process: Ensuring Precision and Reliability
The production of high-quality edta vacuum blood collection tubes is a meticulously controlled process designed to ensure sterility, accurate additive concentration, and robust vacuum integrity. This complex manufacturing chain involves several critical stages, each adhering to strict quality control protocols.
Materials and Components:
- Tube Material: Primarily medical-grade Polyethylene Terephthalate (PET) due to its excellent clarity, break resistance, and gas barrier properties. Glass tubes are also used but are less common now due to safety and disposal concerns.
- Additive: Ethylenediaminetetraacetic acid (EDTA) salts, typically K2EDTA or K3EDTA, used as an anticoagulant. These are precisely dispensed as a liquid solution and then spray-dried onto the inner wall of the tube.
- Stopper: Made from medical-grade synthetic rubber (e.g., bromobutyl or chlorobutyl rubber), ensuring an airtight seal and easy puncture for blood collection. Different colors indicate the additive type.
- Cap: Typically made of polypropylene, providing a protective covering over the stopper.
Production Process Flow:
The manufacturing process for edta blood tube variants generally follows these steps:
1. Tube Formation
Injection molding of PET pellets into precise tube dimensions. Quality checks for consistency in size and wall thickness.
2. Additive Dispensing
Precisely measured liquid EDTA solution is sprayed onto the inner surface of the tube. This ensures even coating and optimal anticoagulant-to-blood ratio.
3. Drying Process
The tubes undergo a controlled drying phase to evaporate solvents, leaving a thin, uniform film of EDTA powder.
4. Stopper & Cap Assembly
Sterile stoppers are precisely inserted, and caps are applied. This critical step forms the vacuum seal.
5. Vacuum Creation & Testing
A precise vacuum is drawn within each tube. Automated systems verify vacuum pressure, ensuring accurate fill volumes during blood collection.
6. Sterilization & Packaging
Tubes are sterilized, typically using gamma irradiation, and then labeled and packaged in sterile conditions. Compliance with ISO 11137 is paramount.
Testing Standards and Quality Assurance:
All stages are subjected to rigorous quality control to meet international standards such as ISO 6710 (Single-use container111s for human venous blood specimen collection), CLSI GP34 (Collection of Diagnostic Venous Blood Specimens), and ISO 13485 (Medical devices – Quality management systems). This ensures optimal performance, consistent vacuum pressure, and precise anticoagulant concentration, critical for the integrity of blood samples. Our EDTA vacuum tube products have a typical service life (shelf life) of 18-24 months from the date of manufacture, provided they are stored under recommended conditions.
Target Industries and Advantages:
These tubes are indispensable in a wide array of sectors, including clinical diagnostic laboratories, blood banks, research institutions, pharmaceutical companies, and veterinary clinics. In typical application scenarios, such as routine complete blood count (CBC) testing, the precise EDTA concentration ensures optimal cell preservation, preventing hemolysis and agglutination, which would otherwise lead to erroneous results. This precision directly translates to energy saving by reducing the need for repeat sample collections and re-tests, while the robust, non-corrosive PET material ensures sample integrity and safety throughout transport and processing.
Technical Specifications and Parameters
Understanding the precise technical specifications of edta vacuum blood collection tubes is fundamental for laboratory professionals to ensure compatibility with analytical instrumentation and adherence to assay requirements. The critical parameters define the tube's performance and suitability for various diagnostic applications.

Image: Typical EDTA and plain tubes for blood collection.
Product Specification Table: EDTA Vacuum Blood Collection Tubes
| Parameter | Specification | Significance |
|---|---|---|
| Tube Material | Medical-grade PET (Polyethylene Terephthalate) | Ensures excellent optical clarity, chemical stability, and robust break resistance, safeguarding samples and personnel. |
| Additive Type | K2EDTA or K3EDTA (Spray-dried) | Ethylenediaminetetraacetic acid chelates calcium ions, effectively preventing blood coagulation. K2EDTA is recommended by CLSI for hematology, offering less erythrocyte shrinkage than K3EDTA. |
| Nominal Fill Volume | 2.0 mL, 3.0 mL, 4.0 mL, 5.0 mL, 6.0 mL (customizable) | Precise fill volume is critical to maintain the correct anticoagulant-to-blood ratio (typically 1:10), preventing dilution or insufficient anticoagulation. |
| Anticoagulant Concentration | 1.8 mg EDTA/mL of blood (CLSI standard for K2EDTA) | Optimized concentration ensures effective anticoagulation without adverse effects on cellular morphology or analytical results. |
| Tube Dimensions | 13x75mm, 13x100mm, 16x100mm (Diameter x Length) | Standardized dimensions ensure compatibility with automated laboratory analyzers and centrifuges. |
| Cap Color | Lavender/Purple (ISO 6710 standard) | Universal color coding for immediate identification of EDTA anticoagulant, reducing pre-analytical errors. |
| Sterilization Method | Gamma Irradiation (10^-6 SAL) | Ensures product sterility, crucial for preventing contamination of blood samples. |
| Shelf Life | 18-24 months from manufacturing date | Indicates stability of vacuum and additive effectiveness under recommended storage conditions. |
| Vacuum Stability | Guaranteed until expiration date | Ensures consistent and accurate fill volumes throughout the product's life, vital for proper blood-to-anticoagulant ratio. |
The selection between K2EDTA and K3EDTA potassium edta tube variants often depends on specific laboratory preferences and instrument manufacturer recommendations. While both effectively chelate calcium, K2EDTA is generally preferred for hematology by organizations like the CLSI due to its minimal impact on red blood cell morphology and smaller effect on packed cell volume (PCV) measurements, as it does not contribute to plasma volume like liquid K3EDTA solution.
Diverse Application Scenarios
The utility of edta vacuum blood collection tubes extends across a broad spectrum of diagnostic and research applications, serving as the primary choice for analyses requiring whole blood or plasma. Their ability to prevent coagulation while preserving cellular integrity makes them indispensable.
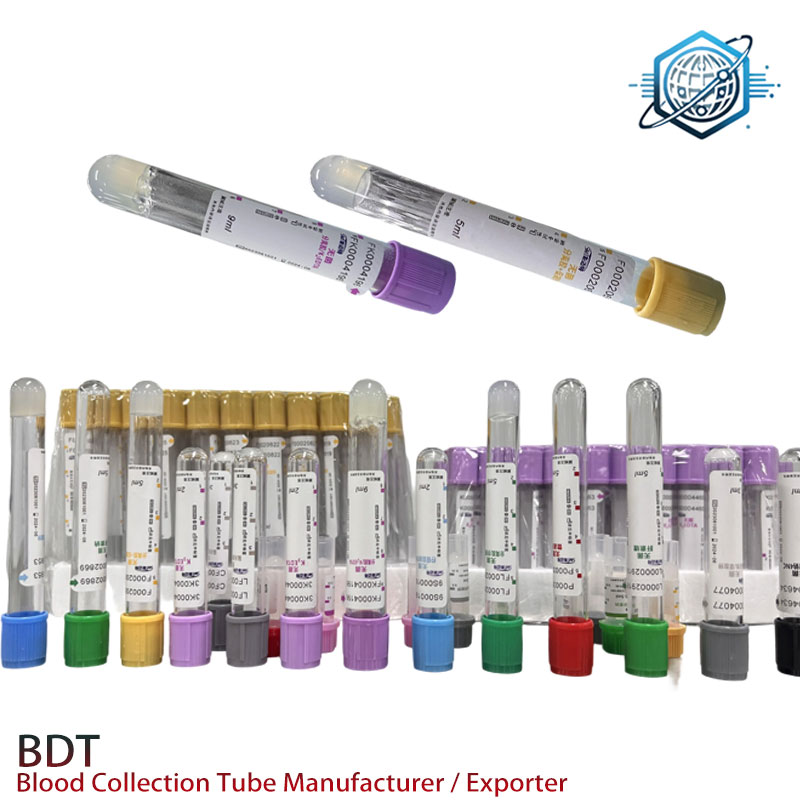
Image: Plain Red Top Tube, often contrasted with EDTA tubes for different tests.
Key Areas of Application:
- Hematology: The most common application. EDTA tubes are essential for tests such as Complete Blood Count (CBC), which measures red blood cells, white blood cells, and platelets; erythrocyte sedimentation rate (ESR); blood typing; and cross-matching. The anticoagulant preserves cell morphology, crucial for accurate cell counting and differentiation.
- Molecular Diagnostics: Used for DNA and RNA extraction from whole blood. EDTA chelates magnesium ions, which are cofactors for many nucleases, thereby protecting nucleic acids from degradation. This is vital for genetic testing, pathogen detection, and oncology research.
- Immunology: Certain immunological tests that require whole blood or plasma for analysis of cytokines, antibodies, or specific cell populations utilize EDTA tubes. It prevents clotting artifacts that could interfere with assay results.
- Blood Banking: For donor screening, compatibility testing, and preparing blood components, EDTA tubes are critical for maintaining sample integrity until processing.
- Flow Cytometry: For precise analysis of cell surface markers and intracellular components, EDTA-anticoagulated blood ensures cell viability and prevents clumping, allowing for accurate cell population identification.
The consistent performance of our edta vacuum tube products in these varied settings underscores their reliability and suitability for high-stakes medical diagnostics. Laboratories rely on the consistent vacuum draw and precise additive concentration to ensure that collected samples are truly representative for subsequent analysis.
Technical Advantages and Performance Excellence
The engineering and design of our edta vacuum blood collection tubes confer several distinct technical advantages, leading to superior sample quality and enhanced laboratory efficiency. These advantages are pivotal for modern diagnostic practices.
Core Advantages:
- Uncompromised Sample Integrity: The precisely spray-dried EDTA ensures optimal anticoagulation without dilution effects observed with liquid EDTA, preserving cellular morphology and preventing platelet aggregation, critical for accurate hematological parameters.
- Consistent Vacuum Draw: Each tube is manufactured with a meticulously controlled vacuum, guaranteeing the collection of the exact specified volume of blood. This maintains the crucial blood-to-anticoagulant ratio, vital for preventing either partial clotting or excessive dilution that could lead to erroneous results.
- Reduced Pre-analytical Errors: The standardized design, clear labeling, and universal cap coloration minimize the risk of miscollection or misidentification of samples, a significant source of errors in laboratory diagnostics.
- Enhanced Safety: Constructed from break-resistant PET, our tubes significantly reduce the risk of breakage during transport and centrifugation compared to glass tubes, thereby improving safety for healthcare personnel and reducing biohazard exposure. The secure stopper design also prevents leakage.
- Compatibility with Automation: Our tubes are designed for seamless integration with automated blood collection, processing, and analysis systems. Their consistent dimensions and robust construction ensure smooth handling by robotic systems, enhancing throughput and reducing manual intervention.
- Extended Shelf Life: Optimized manufacturing processes and material selection contribute to an extended shelf life of 18-24 months, ensuring long-term reliability and inventory management efficiency for laboratories.
- Biocompatibility: All materials used are medical-grade and extensively tested for biocompatibility, ensuring no interference with sample components or diagnostic assays.
These advantages collectively translate into higher diagnostic confidence, operational efficiencies, and improved patient outcomes, solidifying the position of our edta blood tube as a premium choice in clinical settings.
Vendor Comparison and Competitive Edge
In a competitive market dominated by established brands, selecting the right vendor for edta vacuum blood collection tubes involves a detailed evaluation of product quality, consistency, and overall value. While market leaders like BD (e.g., Vacutainer®) and Greiner Bio-One (e.g., Vacuette®) set high benchmarks, our products offer a compelling alternative by focusing on specific differentiators.
Competitive Comparison: Key Parameters
| Feature | Our EDTA Tubes | Major Competitor A (e.g., BD Vacutainer®) | Major Competitor B (e.g., Greiner Bio-One Vacuette®) |
|---|---|---|---|
| Tube Material | Medical-grade PET, high clarity | Medical-grade PET, often with specialized coatings | Medical-grade PET |
| Additive Application | Optimized Spray-Drying for uniform coating | Proprietary spray-drying methods | Advanced spray-drying technology |
| Vacuum Consistency | ±5% variation across batches (rigorously tested) | ±5% (Industry Standard) | ±5% (Industry Standard) |
| Stopper Material/Design | Butyl rubber, self-sealing, low particulate generation | Halobutyl rubber, often with Hemogard™ technology | Synthetic rubber, various safety cap designs |
| Trace Element Leaching | Ultra-low, certified for sensitive assays | Low, generally suitable for most assays | Low, suitable for most assays |
| Cost-Effectiveness | Highly competitive pricing without sacrificing quality | Premium pricing | Premium pricing |
| Customization Options | Extensive for volume, additive, labeling | Limited for standard products | Moderate for large orders |
| Certifications | ISO 13485, CE, FDA-registered | ISO 13485, CE, FDA, numerous national certifications | ISO 13485, CE, FDA, various national certifications |
Our commitment to stringent quality control, coupled with advanced manufacturing techniques, allows us to deliver edta vacuum tube products that consistently meet or exceed industry standards. We distinguish ourselves through a blend of superior product performance, flexible customization capabilities, and a highly competitive pricing structure, offering an excellent value proposition for laboratories seeking reliable and cost-effective blood collection solutions. Our focus on user feedback and continuous improvement also ensures that our products evolve with the dynamic needs of the diagnostic industry, including meeting demand for specialized products like small edta tubes.
Customized Solutions for Specialized Needs
Recognizing that standard products may not always perfectly align with unique laboratory requirements, we offer comprehensive customized solutions for our edta vacuum blood collection tubes. This flexibility is particularly valuable for research institutions, specialized diagnostic centers, and pharmaceutical companies with specific project demands.

Image: Assortment of EDTA and plain tubes illustrating customization potential.
Our Customization Capabilities Include:
- Tailored Fill Volumes: Beyond standard sizes, we can produce tubes with specific nominal fill volumes to accommodate unique blood sample requirements, crucial for pediatric collections or highly sensitive assays.
- Additive Concentration Adjustments: While standard EDTA concentrations adhere to CLSI guidelines, certain research protocols or specialized applications may require specific K2EDTA or K3EDTA concentrations, which we can provide.
- Custom Labeling and Barcoding: For enhanced workflow and inventory management, we offer options for custom printed labels, including specific logos, barcodes, batch numbers, or unique identifiers as per client specifications.
- Special Packaging: Whether it's unique quantities per rack, specialized secondary packaging for automated systems, or specific sterilization indicators, our packaging solutions can be adapted to client needs.
- Small Batch Production: For pilot studies or specialized research, we can accommodate smaller batch orders, providing the same level of quality and precision as our large-scale production.
Our team of technical experts works closely with clients from initial consultation through design and production, ensuring that customized edta vacuum tube solutions meet precise performance criteria and regulatory compliance. This collaborative approach allows us to deliver products that are perfectly aligned with client operational demands and scientific objectives, even for highly specialized vacutainer edta tubes applications.
Application Case Studies: Real-World Impact
The reliability and precision of our edta vacuum blood collection tubes are best demonstrated through their successful application in diverse clinical and research environments. Here are illustrative case studies highlighting their impact:
Case Study 1: Large Academic Medical Center – Enhancing Hematology Workflow
A prominent university hospital faced challenges with inconsistent fill volumes and occasional micro-clot formation when using their previous supplier's EDTA tubes for daily CBC and differential testing, leading to re-draws and delayed results. Upon transitioning to our edta vacuum tube products, they observed a significant improvement. The hospital reported a 15% reduction in re-collection rates due to consistent vacuum pressure and optimal EDTA dispersion, directly improving patient turnaround times and laboratory efficiency. The robust PET construction also reduced tube breakage during pneumatic tube transport, enhancing safety and sample integrity. This led to annual savings of approximately $20,000 in consumables and labor for repeat procedures, showcasing the tangible benefits of reliable pre-analytical components.
Case Study 2: Biotechnology Firm – Precision for Genomic Research
A leading biotechnology firm specializing in pharmacogenomics required exceptionally stable blood samples for large-scale DNA and RNA extraction. They needed tubes that guaranteed minimal degradation of nucleic acids and high purity for downstream next-generation sequencing (NGS) applications. Our potassium edta tube, specifically designed with ultra-low trace element leaching and optimized K2EDTA concentration, proved ideal. The firm noted a consistent yield of high-quality nucleic acids, with an average A260/280 ratio of 1.85-1.90 for DNA, well within optimal ranges for NGS. This consistency was crucial for their high-throughput genomic studies, reducing batch variation and accelerating research milestones. The reliability of our tubes contributed to faster validation of genetic markers for drug response, demonstrating the critical role of pre-analytical quality in advanced research.
Case Study 3: Pediatric Oncology Unit – Safety and Small Volume Accuracy
For a pediatric oncology unit, the challenge was collecting small, precise blood volumes from young patients, minimizing discomfort and ensuring diagnostic accuracy from limited samples. They adopted our customized small edta tubes (1.0 mL fill volume). The small diameter and precise vacuum allowed for accurate, less invasive collection. The unit reported enhanced ease of use for phlebotomists and improved comfort for pediatric patients. More importantly, the exact anticoagulant-to-blood ratio ensured the integrity of precious small volume samples for specialized hematology and molecular tests, eliminating the need for distressing re-draws and contributing to better patient experience and care. This case exemplifies our capability to meet highly specialized clinical needs with targeted product solutions.
Ensuring Trust and Authority: Our Commitment to Quality
Certifications and Compliance:
Our manufacturing facilities and products adhere to the most rigorous international quality and regulatory standards. We are certified under ISO 13485 (Quality Management System for Medical Devices), ensuring a systematic approach to quality control from design to delivery. Our edta vacuum blood collection tubes are CE marked, affirming compliance with European Union health, safety, and environmental protection directives, and are registered with the FDA, meeting stringent U.S. regulatory requirements for medical devices. These certifications underscore our unwavering commitment to producing safe, effective, and reliable diagnostic consumables.
Partnerships and Expertise:
With over 15 years of dedicated service in the medical device industry, we have cultivated enduring partnerships with leading diagnostic laboratories, major hospital networks, and research institutions globally. Our long-standing expertise in blood collection technology is built on a foundation of continuous research and development, collaborating with clinical experts to anticipate and meet evolving diagnostic needs. Our product development cycles incorporate feedback from hundreds of customers, ensuring our edta vacuum tube offerings are validated by real-world usage and performance data.
Logistics, Support, and Guarantees (Trustworthiness)
Frequently Asked Questions (FAQ):
Q: What is the primary difference between K2EDTA and K3EDTA?
A: Both K2EDTA and K3EDTA prevent coagulation by chelating calcium. The Clinical and Laboratory Standards Institute (CLSI) recommends K2EDTA for hematology as it causes less erythrocyte shrinkage and has a smaller impact on cell morphology and packed cell volume compared to K3EDTA, particularly in liquid form, which can dilute the sample. Our potassium edta tube products are available in both forms.
Q: How can I ensure the correct fill volume for accurate results?
A: Our edta vacuum blood collection tubes are designed with a precise vacuum to draw the specified volume of blood. Always allow the tube to fill completely until the blood flow stops naturally. Under-filling or over-filling can alter the blood-to-anticoagulant ratio, potentially leading to inaccurate results.
Q: Are your tubes compatible with automated laboratory analyzers?
A: Yes, our tubes are manufactured to industry-standard dimensions and specifications (e.g., 13x75mm, 13x100mm) to ensure seamless compatibility with most automated hematology analyzers and pre-analytical systems, including those designed for vacutainer edta tubes.
Q: What is the typical lead time for large orders?
A: Standard orders for our edta blood tube products typically have a lead time of 2-4 weeks, depending on order size and destination. For customized solutions or exceptionally large volumes, lead times may vary and will be confirmed at the time of order placement. We maintain robust inventory levels and an efficient logistics network to ensure timely fulfillment.
Lead Time and Fulfillment:
We are committed to efficient order processing and delivery. Standard product orders are typically fulfilled within 14-28 business days from confirmation. For bulk orders or specific customized requests, our dedicated account managers will provide a detailed production and shipping schedule. Our global distribution network ensures that products reach our clients efficiently and in optimal condition, respecting cold chain requirements where applicable.
Warranty and After-Sales Support:
All our edta vacuum blood collection tubes come with a comprehensive product warranty covering manufacturing defects and performance parameters for their stated shelf life. Our commitment extends beyond delivery, with a responsive after-sales support team ready to assist with any technical inquiries, product performance issues, or logistics concerns. We provide detailed technical documentation and training resources to ensure optimal product usage. Our goal is to forge long-term partnerships built on trust and mutual success.
References
- Clinical and Laboratory Standards Institute. (2017). Procedures for the Collection of Diagnostic Blood Specimens by Venipuncture; Approved Standard—Seventh Edition. CLSI document GP34-A7. Wayne, PA: CLSI.
- International Organization for Standardization. (2010). ISO 6710: Single-use container111s for human venous blood specimen collection. Geneva, Switzerland: ISO.
- International Organization for Standardization. (2016). ISO 13485: Medical devices – Quality management systems – Requirements for regulatory purposes. Geneva, Switzerland: ISO.
- International Organization for Standardization. (2017). ISO 11137-1: Sterilization of health care products – Radiation – Part 1: Requirements for development, validation and routine control of a sterilization process for medical devices. Geneva, Switzerland: ISO.
- Lippi, G., & Plebani, M. (2016). The IFCC Academy Project. The importance of the pre-analytical phase. eJIFCC, 27(2), 177-192.
-
Deluxe Electric Foot Warmer With Zipper-Shijiazhuang BDT Heating Pad Co.,Ltd.|Fast Heating,Customizable Design
NewsSep.02,2025 -
Deluxe Electric Foot Warmer- Shijiazhuang BDT Heating Pad Co.,Ltd.|Fast Heating,Zipper Design
NewsSep.02,2025 -
Deluxe Electric Foot Warmer- Shijiazhuang BDT Heating Pad Co., Ltd.|Fast Heating, Custom Fit
NewsSep.02,2025 -
Deluxe Electric Foot Warmer With Zipper - Shijiazhuang BDT Heating Pad Co., Ltd.
NewsSep.02,2025 -
Fast Heating Technology Deluxe Electric Foot Warmer - Shijiazhuang BDT Heating Pad Co., Ltd.
NewsSep.02,2025 -
Deluxe Electric Foot Warmer-Fast Heating Technology|Shijiazhuang BDT Heating Pad Co., Ltd.
NewsSep.02,2025














