- bdttonyjiao@hotmail.com
- +86 +8615081115902
- 9-1-701 TonfuTown Shijiahzuang City Hebei Province China
Aug . 31, 2025 04:40 Back to list
Accurate Coagulation Tests: Sodium Citrate Tube Used For Blood Collection
Understanding the Critical Role of Sodium Citrate Tubes in Clinical Diagnostics
In the demanding landscape of modern clinical diagnostics, precision and sample integrity are paramount. Among the array of blood collection devices, the sodium citrate tube used for coagulation studies stands as a cornerstone. These specialized tubes are meticulously designed to ensure accurate assessment of blood clotting mechanisms, which is vital for diagnosing a wide range of conditions, from bleeding disorders to thrombotic risks. The precise formulation and manufacturing of these tubes directly impact the reliability of patient results, influencing treatment decisions and patient safety. Our Sodium Citrate 1:4 Tube, for instance, represents a commitment to these high standards, offering a reliable solution for critical diagnostic needs.
The primary function of the sodium citrate anticoagulant is to bind calcium ions in the blood, effectively preventing coagulation without significantly altering cellular components. This makes it indispensable for preserving the integrity of platelets and plasma proteins involved in the coagulation cascade. As we delve deeper, we will explore the intricate manufacturing processes, technical specifications, and broad application scenarios that underscore the importance of this essential diagnostic tool, ensuring that the collected sodium citrate whole blood sample remains viable for accurate analysis.
Industry Trends and Market Dynamics for Blood Collection Systems
The global market for blood collection tubes, including the specialized citrate test tube, is experiencing steady growth, driven by an increasing prevalence of chronic diseases, a rising number of surgical procedures, and advancements in diagnostic technologies. Key trends influencing this sector include:
- Automation and Standardization: There's a continuous push towards automated pre-analytical systems in laboratories, requiring tubes with consistent dimensions and anticoagulant concentrations for seamless integration.
- Enhanced Safety Features: Development of safety-engineered blood collection devices, such as tubes with safety closures and needle-stick prevention features, is a priority to protect healthcare workers.
- Material Innovations: Research into new, inert materials for tubes that minimize sample contamination and interaction with anticoagulants, extending sample stability.
- Diagnostic Expansion: The growth of personalized medicine and specialized diagnostics, particularly in areas like oncology and genetic testing, increases the demand for highly reliable sample collection.
- Sustainability: Increasing focus on eco-friendly manufacturing processes and recyclable materials for blood collection tubes to reduce environmental impact.
These trends collectively underscore the critical need for high-quality, reliable, and compliant blood collection tubes. The demand for precise sodium citrate for blood collection will only intensify as diagnostic capabilities expand and patient outcomes become increasingly dependent on accurate initial sample integrity.
Technical Specifications and Material Science of the Sodium Citrate Tube
A deep understanding of the technical specifications and material properties is crucial for appreciating the reliability of the sodium citrate tube used for coagulation testing. These tubes are not merely container111s; they are sophisticated medical devices engineered for precision.
Core Components and Materials:
- Tube Material: Typically made from medical-grade polyethylene terephthalate (PET), known for its excellent barrier properties against gas exchange and chemical inertness. Glass tubes are also available but PET offers superior shatter resistance and lighter weight.
- Cap Material: Often made from polypropylene or rubber, designed for a secure, leak-proof seal and ease of puncturing by automated analyzers without coreing.
- Anticoagulant: Trisodium citrate solution (typically 3.2% or 3.8% w/v), precisely formulated to a blood-to-anticoagulant ratio of 9:1 or 1:4 (as with our product) to ensure optimal calcium chelation for coagulation tests. The exact ratio is critical to avoid dilution effects or incomplete anticoagulation.
- Lubricant/Coating: Some tubes may feature a silicone coating on the inner surface to prevent cell adhesion and hemolysis, preserving sample quality.
Product Specification Table: Sodium Citrate 1:4 Tube
| Parameter | Specification | Rationale/Benefit |
|---|---|---|
| Anticoagulant | Trisodium Citrate, 3.2% w/v | Standard concentration recommended by CLSI for coagulation tests. |
| Blood-to-Anticoagulant Ratio | 1:4 (Blood:Citrate) | Optimized for specific dilution-sensitive assays, e.g., erythrocyte sedimentation rate (ESR). |
| Tube Material | Medical-grade PET | Chemical inertness, optical clarity, reduced gas permeability. |
| Tube Dimensions (Typical) | 13 x 75 mm or 13 x 100 mm | Compatibility with automated blood analyzers and centrifuges. |
| Draw Volume (Typical) | 2.7 mL to 5.0 mL | Accommodates various test requirements and patient sample volumes. |
| Sterilization Method | Gamma Irradiation | Ensures sterility without compromising material integrity or anticoagulant efficacy. |
| Shelf Life | 18-24 Months from manufacturing date | Long-term usability and inventory management for laboratories. |
The Manufacturing Process of a High-Quality Sodium Citrate Tube
The production of a reliable sodium citrate tube used for critical diagnostics is a sophisticated multi-stage process, demanding stringent quality control and adherence to international standards. This intricate process ensures that each tube consistently delivers the performance required for accurate diagnostic results.
Schematic Manufacturing Steps:
-
Raw Material Sourcing & Inspection:
High-purity medical-grade PET granules and pharmaceutical-grade trisodium citrate are sourced from approved suppliers. Incoming materials undergo rigorous quality control (QC) checks, including spectroscopic analysis for purity and rheological testing for PET. Adherence to USP/EP standards for chemicals and ISO 10993 for biocompatibility of plastics.
-
Tube Molding (Injection Molding):
PET granules are melted and injected into precision molds under controlled temperature and pressure to form the tube body. This process ensures consistent tube dimensions (e.g., ISO 6710 compliance for external dimensions) and wall thickness, critical for automated handling and centrifugation. The caps are also molded, often from polypropylene, designed for optimal sealing and piercing.
-
Anticoagulant Preparation & Dispensing:
Trisodium citrate solution is prepared in a sterile environment using deionized water and pharmaceutical-grade sodium citrate, precisely measured to achieve 3.2% or 3.8% w/v concentration. Automated, high-precision dispensing systems then add the exact volume of anticoagulant to each tube. This volumetric accuracy is paramount for maintaining the correct blood-to-anticoagulant ratio (e.g., 9:1 or 1:4), which directly impacts coagulation test results.
-
Drying/Coating (Optional):
For certain applications or to enhance mixing, the anticoagulant may be spray-dried onto the inner wall of the tube. Some manufacturers apply an inert silicone coating to the inner surface to minimize interaction with blood cells and reduce hemolysis.
-
Cap Assembly & Vacuum Sealing:
Caps are robotically placed onto the tubes. A crucial step involves creating a precise vacuum within the tube. This vacuum is measured and controlled to ensure the correct blood draw volume when used with venipuncture needles, adhering to standards like CLSI GP39-A6.
-
Sterilization:
The assembled tubes are sterilized, most commonly using gamma irradiation. This method ensures a sterility assurance level (SAL) of 10-6 (e.g., ISO 11137 compliance), effectively eliminating microorganisms without degrading the PET or the sodium citrate. Post-sterilization, a biological indicator test is performed to validate the process.
-
Final Inspection & Packaging:
Each tube undergoes automated visual inspection for defects, particulate matter, and correct fill volume. Labels are applied with lot numbers and expiry dates. Tubes are then packaged into trays and boxes within a cleanroom environment, ready for distribution.
Throughout this process, continuous in-process quality control checks are performed, including dimensional measurements, vacuum retention tests, leak tests, and pH checks of the anticoagulant. The entire manufacturing facility operates under ISO 13485 (Medical Devices Quality Management System) certification, ensuring traceability and consistency from raw material to finished product. The typical service life for these tubes is between 18 to 24 months, contingent on proper storage conditions.
Application Scenarios and Target Industries
The versatility of the sodium citrate whole blood sample obtained using these tubes makes them indispensable across a spectrum of medical and research applications. Their primary use is in the collection of samples for coagulation studies, but their utility extends beyond.
Primary Diagnostic Applications:
- Prothrombin Time (PT): Measures the extrinsic and common pathways of coagulation, crucial for monitoring warfarin therapy and assessing liver function.
- Activated Partial Thromboplastin Time (aPTT): Evaluates the intrinsic and common pathways, essential for monitoring heparin therapy and detecting intrinsic factor deficiencies.
- Fibrinogen Levels: Quantifies the primary substrate for clot formation, important in inflammatory conditions, bleeding, and thrombotic risk assessment.
- D-dimer: Used to rule out venous thromboembolism (VTE) such as deep vein thrombosis (DVT) and pulmonary embolism (PE), as well as to monitor disseminated intravascular coagulation (DIC).
- ESR (Erythrocyte Sedimentation Rate): Although not a coagulation test, the 1:4 sodium citrate tube is specifically designed for ESR measurements, aiding in the diagnosis of inflammatory conditions.
- Special Coagulation Factors: Assays for specific factors like Factor VIII, Factor IX, or von Willebrand Factor.
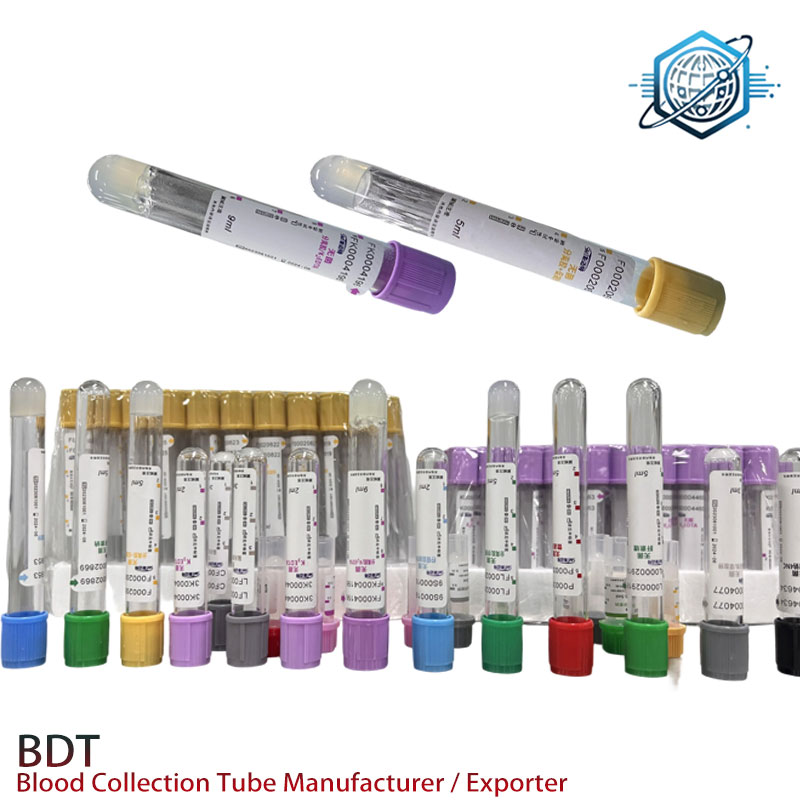
Target Industries and Advantages:
- Clinical Laboratories: The primary end-users, requiring high-volume, reliable tubes for routine and specialized coagulation panels. Advantages include consistent quality for precise results and compatibility with automated systems.
- Hospitals & Blood Banks: For patient diagnostics, pre-surgical screening, and donor screening. Key advantages are ensuring sample integrity and safety, critical for transfusion medicine.
- Pharmaceutical & Biotech Research: In drug development, particularly for anticoagulants or antiplatelet agents, accurate coagulation assays are vital. The tubes provide a standardized method for sample collection in clinical trials.
- Academic Research Institutions: For studying thrombosis, hemostasis, and various blood disorders. The precise blood-to-anticoagulant ratio is essential for experimental reproducibility.
The ability of a quality sodium citrate for blood collection tube to maintain sample integrity for extended periods offers significant advantages, including reduced pre-analytical errors, improved diagnostic accuracy, and ultimately, better patient care outcomes.
Technical Advantages and Performance Metrics
The technical advantages of a superior citrate test tube are manifold, directly contributing to the accuracy and reliability of diagnostic results. These advantages stem from meticulous design, material selection, and manufacturing precision.
Key Technical Advantages:
- Precision Anticoagulant Ratio: The exact 9:1 or 1:4 (blood:anticoagulant) ratio is critical. Deviations can lead to erroneous results (e.g., falsely prolonged PT/aPTT due to over-dilution, or clot formation due to under-dilution). Advanced manufacturing ensures this ratio is maintained within tight tolerances (e.g., ±5%).
- Vacuum Consistency: A precisely controlled vacuum ensures the correct volume of blood is drawn, preventing under-filling (which distorts the blood:anticoagulant ratio) or over-filling. Modern tubes boast vacuum consistency within ±10% of stated draw volume.
- Inert Materials: Use of medical-grade PET plastic prevents interaction between the tube material and blood components, ensuring the stability of coagulation factors. This minimizes activation of platelets or coagulation cascade prior to testing.
- Optimal pH Stability: The sodium citrate solution is buffered to maintain an optimal pH range for preserving coagulation factor activity for up to 24 hours post-collection at room temperature, as per CLSI guidelines.
- Leak-Proof & Evaporation Resistance: High-quality stoppers ensure sample integrity during transport and storage, preventing leakage and minimizing evaporation of the anticoagulant, which could alter its concentration.
- Compatibility with Automation: Standardized dimensions and cap designs ensure seamless integration with automated laboratory systems for centrifugation, uncapping, and analysis, improving lab efficiency and reducing manual errors.
Performance Metrics (Example Data):
- CV for Anticoagulant Volume: < 2% (Industry best practice)
- Vacuum Retention Rate: > 95% over 1 year (Critical for shelf life and reliable draw volume)
- Hemolysis Index: < 0.5% (Ensures sample quality for assays sensitive to red cell lysis)
- Platelet Activation Markers (e.g., P-selectin): No significant increase over baseline within 4 hours (Indicates minimal pre-analytical activation).
Vendor Comparison: Key Differentiators in Sodium Citrate Tubes
When selecting a supplier for the sodium citrate tube used for critical diagnostic work, understanding the differentiating factors between vendors is paramount. While core specifications may appear similar, subtle differences in manufacturing, quality control, and innovation can significantly impact performance and reliability.
Product Comparison Table: Leading Sodium Citrate Tube Features
| Feature/Metric | High-Quality Vendor (e.g., Our Sodium Citrate 1:4 Tube) | Standard/Economical Vendor |
|---|---|---|
| Anticoagulant Purity | Pharmaceutical Grade (USP/EP), Lot-specific CoA | Technical Grade, general CoA |
| Anticoagulant Volume Accuracy | CV < 2%, Gravimetric dispensing with online QC | CV 2-5%, Volumetric dispensing, batch QC |
| Vacuum Stability/Shelf Life | 24 months, >98% vacuum retention at expiry | 12-18 months, 90-95% vacuum retention |
| Tube Material Quality | Virgin Medical-Grade PET, low extractables | Standard PET, potential for higher extractables |
| Stopper Design | Self-sealing, low-coring, synthetic rubber for chemical resistance | Basic rubber, higher risk of coring or leakage |
| Certifications | ISO 13485, CE Mark, FDA 510(k) cleared | Basic ISO 9001, local approvals |
| Packaging | Robust, humidity-controlled, clear traceability | Standard, basic protection |
Customized Solutions and Client Partnerships
Recognizing that every laboratory and clinical setting may have unique requirements, leading manufacturers of the sodium citrate for blood collection offer customized solutions. This flexibility ensures that specific operational needs, regional standards, or specialized research demands are met with precision.
Areas for Customization:
- Tube Dimensions & Draw Volume: While standard sizes exist, some labs require specific tube lengths or diameters for compatibility with unique automated systems or reduced draw volumes for pediatric/geriatric patients.
- Anticoagulant Concentration/Ratio: Although 3.2% w/v is standard, certain research applications or older instruments might necessitate 3.8% w/v or a different blood-to-anticoagulant ratio.
- Labeling & Barcoding: Custom barcodes (e.g., 1D, 2D), specific information printing, or color-coding on labels to integrate seamlessly with a facility’s LIS (Laboratory Information System).
- Packaging Configurations: Custom tray sizes, sterile blister packs, or specific unit quantities per box to optimize workflow and storage.
- Specialty Additives: For highly specialized research, inert coatings or other minor additives can be incorporated if compatible with the base tube and anticoagulant.

Our Approach to Partnership:
We believe in fostering long-term partnerships. Our dedicated technical support and R&D teams work closely with clients to understand their specific challenges and develop tailored solutions. This collaborative approach ensures that our products, including the specialized sodium citrate tube used for critical applications, not only meet but exceed expectations, contributing to enhanced diagnostic efficiency and reliability. Our years of service in the medical device industry have equipped us with the expertise to navigate complex client requirements, from large hospital networks to specialized research facilities.
Application Case Studies and Customer Experience
Real-world application demonstrates the tangible benefits of high-quality blood collection tubes. Here are illustrative case studies highlighting the impact of reliable citrate test tube performance.
Case Study 1: Large Academic Medical Center - Reducing Pre-Analytical Errors
Challenge: A major university hospital laboratory was experiencing a 3% rejection rate for coagulation samples due to incorrect fill volumes and microclots, leading to delayed diagnoses and increased costs from redraws.
Solution: After a comprehensive review, they transitioned to our Sodium Citrate 1:4 Tubes, specifically selecting our high-precision vacuum-controlled tubes.
Outcome: Within six months, the coagulation sample rejection rate dropped to under 0.5%. The consistent vacuum ensured accurate blood draw volumes, and the inert tube material, combined with precise anticoagulant dispensing, virtually eliminated microclot formation. The lab reported a 15% improvement in turnaround time for coagulation tests and significant cost savings.
Customer Feedback: "The consistency of the BDTheatingpads.com sodium citrate tubes has been a game-changer for our coagulation lab. Our technicians trust the product, and our clinicians receive more reliable results faster." - Dr. Emily Chen, Head of Hematology, University Hospital.
Case Study 2: Regional Reference Laboratory - Enhancing Automation Compatibility
Challenge: A large regional reference lab, processing thousands of coagulation samples daily, struggled with tube compatibility issues with their new fully automated analytical platform. Minor dimensional inconsistencies and stopper variations from their previous supplier led to frequent jamming and erroneous processing.
Solution: We provided customized tube dimensions and stopper materials, precisely engineered to the specifications of their automated system. Our tubes underwent rigorous testing for automated loading, uncapping, and sample aspiration compatibility.
Outcome: The lab achieved near-100% compatibility with their automation, drastically reducing manual intervention and system downtime. Throughput increased by 20%, and labor costs for pre-analytical processing were cut by 10%.
Service Case Details: Our technical team conducted on-site assessments and collaborated with the automation vendor to fine-tune tube specifications. This involved several iterations of prototype tubes and extensive validation runs.
Quality Assurance, Certifications, and Authoritative Standards
Authoritativeness and trustworthiness in medical diagnostics are built upon unwavering commitment to quality. Our manufacturing processes for the sodium citrate tube used for critical diagnostics are underpinned by a robust quality management system and adherence to global standards.
Certifications & Compliance:
- ISO 13485:2016 Certified: Our entire quality management system is certified to the international standard for medical devices, ensuring consistency, safety, and effectiveness from design to delivery.
- CE Mark (MDD 93/42/EEC & MDR 2017/745): Our products conform to the essential health and safety requirements for medical devices sold within the European Economic Area.
- FDA 510(k) Cleared: Many of our products, including blood collection tubes, have received market clearance from the U.S. Food and Drug Administration, indicating substantial equivalence to legally marketed predicate devices.
- CLSI Guidelines Adherence: We strictly follow Clinical and Laboratory Standards Institute (CLSI) guidelines, such as GP39-A6 (Tubes and Additives for Venous Blood Specimen Collection), ensuring optimal performance for diagnostic testing.
- USP/EP Standards: Raw materials, particularly trisodium citrate, meet the stringent requirements of the United States Pharmacopeia (USP) and European Pharmacopoeia (EP) for purity and quality.
Quality Control & Test Data:
Each production lot undergoes comprehensive testing, including:
- Anticoagulant Concentration Verification: High-performance liquid chromatography (HPLC) ensures the precise concentration of sodium citrate.
- Vacuum Draw Volume Accuracy: Tested with a blood simulant, guaranteeing accurate fill volumes.
- Leakage & Breakage Tests: Tubes are subjected to pressure and drop tests to ensure structural integrity.
- Sterility Testing: Confirmed through biological indicator assays post-irradiation.
- Coagulation Parameter Validation: Batch samples are tested with reference plasma to confirm no interference with PT, aPTT, and fibrinogen assays.
Our commitment to over two decades of service in this specialized field, coupled with strategic partnerships with leading diagnostic companies globally, further solidifies our authoritative position.
Trustworthiness: FAQ, Lead Time, Warranty & Support
Building trust with B2B clients goes beyond product quality; it encompasses transparency, reliable logistics, and unwavering support.
Frequently Asked Questions (FAQ):
A: The 1:4 sodium citrate tube, such as our Sodium Citrate 1:4 Tube, is primarily sodium citrate tube used for Westergren ESR (Erythrocyte Sedimentation Rate) tests. While 9:1 ratio tubes are for general coagulation studies, the 1:4 ratio is specifically designed for ESR, providing the optimal dilution for accurate erythrocyte sedimentation.
A: Samples should be gently mixed immediately after collection and stored at room temperature (18-25°C). For coagulation tests, plasma should be separated and tested within 4 hours, or stored frozen at -20°C for up to 2 weeks, or at -70°C for longer periods. ESR samples are typically tested within 4 hours at room temperature.
A: Our sodium citrate tubes typically have a shelf life of 18-24 months from the manufacturing date, provided they are stored according to recommended conditions (dry, ambient temperature, away from direct sunlight).
A: Yes, our tubes are designed with standard dimensions (e.g., 13x75mm, 13x100mm) and cap configurations to ensure broad compatibility with leading automated blood coagulation and hematology analyzers. We can provide technical specifications for specific instrument integration.
Lead Time & Fulfillment:
Our streamlined manufacturing and logistics ensure efficient order fulfillment.
- Standard Orders: Typically 3-4 weeks from order confirmation to dispatch, depending on volume.
- Large Volume/Custom Orders: Lead times will be quoted individually based on customization complexity and production schedule, usually ranging from 6-8 weeks.
- Expedited Shipping: Available upon request for urgent requirements, with associated surcharges.
- Global Distribution Network: We utilize a robust global supply chain to ensure timely delivery to various international markets.
Warranty & Customer Support:
We stand by the quality of our products with a comprehensive warranty and dedicated support.
- Product Warranty: All our sodium citrate tubes are warranted against defects in material and workmanship for the stated shelf life under proper storage conditions.
- Technical Support: Our team of highly trained specialists is available to assist with product inquiries, application guidance, troubleshooting, and LIS integration. Support is available via phone, email, and live chat during business hours.
- After-Sales Service: We are committed to prompt investigation and resolution of any product-related concerns. Our complaint handling process is ISO 13485 compliant, ensuring thorough root cause analysis and corrective actions.
Conclusion: The Enduring Importance of the Sodium Citrate Tube
The sodium citrate tube used for coagulation and ESR studies remains a cornerstone in clinical diagnostics. Its meticulous design, precision manufacturing, and adherence to stringent quality standards are critical for ensuring the integrity of blood samples and, consequently, the accuracy of patient diagnoses. As the healthcare landscape continues to evolve, driven by demands for greater efficiency, safety, and diagnostic precision, the role of high-quality blood collection systems becomes even more pronounced.
By understanding the technical intricacies, industry trends, and the commitment to quality from reputable manufacturers, B2B decision-makers can make informed choices that directly impact laboratory performance and, ultimately, patient outcomes. Our dedication to innovation, adherence to global standards, and customer-centric approach ensure that our Sodium Citrate 1:4 Tube and other blood collection solutions provide the reliability and performance expected by modern clinical laboratories and research institutions.

References
- Clinical and Laboratory Standards Institute. Collection of Diagnostic Venous Blood Specimens. CLSI guideline GP41-A7. Wayne, PA: Clinical and Laboratory Standards Institute; 2017.
- Clinical and Laboratory Standards Institute. Tubes and Additives for Venous Blood Specimen Collection; Approved Standard—Sixth Edition. CLSI document GP39-A6. Wayne, PA: Clinical and Laboratory Standards Institute; 2015.
- European Committee for Standardization. EN ISO 13485: Medical devices — Quality management systems — Requirements for regulatory purposes. Geneva, Switzerland: ISO; 2016.
- International Organization for Standardization. ISO 6710: Single-use container111s for human venous blood specimen collection. Geneva, Switzerland: ISO; 2017.
- U.S. Food and Drug Administration. Guidance for Industry and FDA Staff: Blood Establishment Computer System Validation in the Blood Bank and Transfusion Service. Silver Spring, MD: FDA; 2011.
-
Deluxe Electric Foot Warmer With Zipper-Shijiazhuang BDT Heating Pad Co.,Ltd.|Fast Heating,Customizable Design
NewsSep.02,2025 -
Deluxe Electric Foot Warmer- Shijiazhuang BDT Heating Pad Co.,Ltd.|Fast Heating,Zipper Design
NewsSep.02,2025 -
Deluxe Electric Foot Warmer- Shijiazhuang BDT Heating Pad Co., Ltd.|Fast Heating, Custom Fit
NewsSep.02,2025 -
Deluxe Electric Foot Warmer With Zipper - Shijiazhuang BDT Heating Pad Co., Ltd.
NewsSep.02,2025 -
Fast Heating Technology Deluxe Electric Foot Warmer - Shijiazhuang BDT Heating Pad Co., Ltd.
NewsSep.02,2025 -
Deluxe Electric Foot Warmer-Fast Heating Technology|Shijiazhuang BDT Heating Pad Co., Ltd.
NewsSep.02,2025














