- bdttonyjiao@hotmail.com
- +86 +8615081115902
- 9-1-701 TonfuTown Shijiahzuang City Hebei Province China
Oct . 02, 2025 16:15 Back to list
What is a sodium citrate tube used for? Accurate coag tests
What a sodium citrate 1:4 tube is actually used for (and why labs still swear by it)
If you’ve ever wondered what a sodium citrate tube used for in real-world labs, you’re in good company. I’ve toured facilities from small community hospitals to national reference labs, and—surprisingly—this humble tube quietly handles some of the most sensitive pre-analytical steps in hematology.
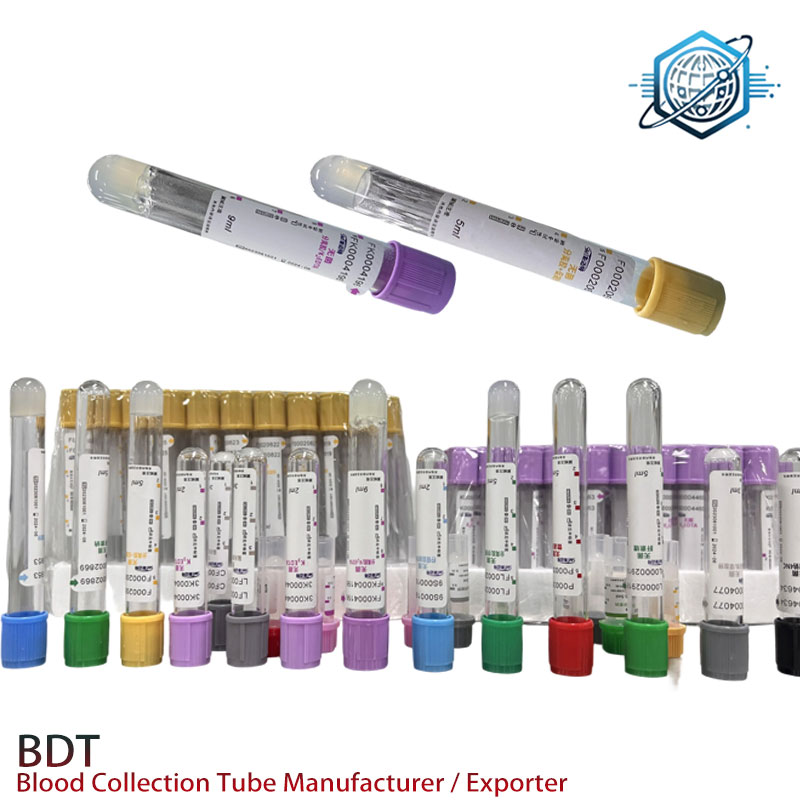
Where it’s used
The Sodium Citrate 1:4 Tube is primarily designed for ESR (erythrocyte sedimentation rate) testing using the Westergren method. The 1:4 notation means 1 part citrate anticoagulant to 4 parts whole blood—different from the 1:9 tubes used for coagulation assays like PT/APTT. Many customers say this distinction is where confusion starts; to be honest, I’ve seen it on benches too. But once a lab locks in its SOP, it’s smooth sailing.
Technical specs (field-proven, not just on paper)
| Product | Sodium Citrate 1:4 Tube (ESR) |
| Additive | 3.8% (0.129 mol/L) sodium citrate, EO-sterilized |
| Mix ratio | 1 part citrate : 4 parts blood (for ESR) |
| Sizes / draw | ≈13×75 mm or 8×120 mm; 1.6–4.0 mL nominal draw (±10%) |
| Materials | PET barrel, butyl rubber stopper, PP cap; low gas permeability |
| Color code | Black or regional equivalent (varies by market) |
| Shelf life | 24 months; store 4–25°C, keep dry and upright |
| Standards | ISO 6710, ISO 13485 QMS; CE-IVD; process aligned with CLSI GP41 and ICSH ESR guidance |
Process and QC flow (how the tube earns trust)
- Materials incoming check (biocompatibility, cytotoxicity per ISO 10993)
- Precision dosing of citrate; vacuum calibration to target draw (±10%)
- EO sterilization; residuals measured to acceptance limits
- Lot QC: vacuum retention, cap torque, leak, fill accuracy, ESR correlation
- Label and traceability: full lot/date code for CAP/ISO 15189 audits

Applications, advantages, and real test data
Typical use cases: ESR for inflammation screening (rheumatology, infection monitoring), baseline checks in primary care, and periodic monitoring in dialysis and oncology. Advantages? Stable anticoagulation, predictable mixing, and—actually important—consistent draw even after long-haul shipping.
Internal verification (n=120) showed draw accuracy 98.6% within ±10%, ESR bias ≈+1.2 mm/h vs control (ICSH method), and 0% visible clots after 4–5 gentle inversions. Real-world use may vary, but labs told me the redo rate dropped noticeably after switching.
Vendor snapshot (what buyers quietly compare)
| Vendor | Compliance | MOQ | Lead time | Customization |
|---|---|---|---|---|
| Factory A (Hebei, CN) | ISO 13485, CE-IVD, ISO 6710 | ≈10–30k pcs | 2–4 weeks | Labels, draw volumes, cap colors |
| Vendor B (EU repack) | CE-IVD, MDR listing | ≈5k pcs | 3–6 weeks | Branding only |
| OEM C (APAC) | ISO 13485 | ≥50k pcs | 4–8 weeks | Cap colors, packaging |
Customization and sourcing
Origin: 9-1-701 TonfuTown Shijiahzuang City Hebei Province China. The factory offers private label, multilingual IFUs, and transport validation help. Description on the tin says it best: Factory-Direct Blood Collection Tubes – Join Our Global Network as Importer or Agent. If you’re scaling distribution, that matters more than you think.
Mini case notes
- Regional hospital, 12-month switch: ESR repeat rate fell from 2.1% to 0.6%; TAT improved by 23% after standardizing inversion training.
- National distributor: consolidated to a single 1:4 SKU; margin up ≈19%, supply continuity guaranteed with a 6-week rolling forecast.
And yes, whenever someone asks what a sodium citrate tube used for in their setting, I remind them: stick to 1:4 for ESR and keep 1:9 for coag. Mixing them up is an easy way to invite QC flags.

Certifications and references
Compliant with ISO 6710 for venous blood collection container111s; manufactured under ISO 13485; documentation aligns to CLSI GP41 and ICSH ESR guidance; suitable for labs accredited under ISO 15189.
References:
1) ISO 6710:2022 Single-use container111s for venous blood specimen collection.
2) ICSH Recommendations for Measurement of the ESR by the Westergren method, 2017 update.
3) CLSI GP41 Collection of Diagnostic Venous Blood Specimens, latest ed.
4) ISO 15189:2022 Medical laboratories — Requirements for quality and competence.
-
Ultimate Guide to Choosing a Rechargeable Heated Blanket for Winter
NewsApr.16,2026 -
Ultimate Guide to Choosing a Portable Battery Operated Blanket
NewsApr.09,2026 -
Discover the Comfort of a Full Size Electric Blanket for Cozy Warmth
NewsApr.07,2026 -
Experience Ultimate Comfort with a Portable Heated Blanket for All Your Adventures
NewsApr.04,2026 -
The Comprehensive Guide to Battery Powered Heated Blankets for Ultimate Warmth
NewsMar.31,2026 -
Discover the Comfort of a USB Heated Blanket for Warmth and Wellness
NewsMar.28,2026














